Acetylcholinesterase
From Proteopedia
(Undo revision 1143776 by David Canner (Talk)) |
|||
| Line 13: | Line 13: | ||
in 1991 opened up new horizons in research on an [http://en.wikipedia.org/wiki/Enzyme enzyme] that had already been the subject of intensive investigation.<ref>PMID:1678899</ref> The unanticipated structure of this extremely rapid enzyme, in which the [http://en.wikipedia.org/wiki/Active_site active site] was found to be buried at the bottom of a <scene name='2ace/Active_site/3'>deep and narrow gorge</scene>, lined by <scene name='2ace/Active_site/4'>14 aromatic residues</scene> <font color='darkmagenta'><b>(colored dark magenta)</b></font>, led to a revision of the views then held concerning [http://en.wikipedia.org/wiki/Substrate_(biochemistry) substrate] traffic, recognition and hydrolysis.<ref>PMID:10545346</ref> To understand how those aromatic residues behave with the enzyme, see [[Flexibility of aromatic residues in acetylcholinesterase]]. Solution of the 3D structure of acetylcholinesterase led to a series of theoretical and experimental studies, which took advantage of recent advances in theoretical techniques for treatment of [http://en.wikipedia.org/wiki/Protein proteins], such as | in 1991 opened up new horizons in research on an [http://en.wikipedia.org/wiki/Enzyme enzyme] that had already been the subject of intensive investigation.<ref>PMID:1678899</ref> The unanticipated structure of this extremely rapid enzyme, in which the [http://en.wikipedia.org/wiki/Active_site active site] was found to be buried at the bottom of a <scene name='2ace/Active_site/3'>deep and narrow gorge</scene>, lined by <scene name='2ace/Active_site/4'>14 aromatic residues</scene> <font color='darkmagenta'><b>(colored dark magenta)</b></font>, led to a revision of the views then held concerning [http://en.wikipedia.org/wiki/Substrate_(biochemistry) substrate] traffic, recognition and hydrolysis.<ref>PMID:10545346</ref> To understand how those aromatic residues behave with the enzyme, see [[Flexibility of aromatic residues in acetylcholinesterase]]. Solution of the 3D structure of acetylcholinesterase led to a series of theoretical and experimental studies, which took advantage of recent advances in theoretical techniques for treatment of [http://en.wikipedia.org/wiki/Protein proteins], such as | ||
[http://en.wikipedia.org/wiki/Molecular_dynamics molecular dynamics] and [http://en.wikipedia.org/wiki/Electrostatics electrostatics] and to [http://en.wikipedia.org/wiki/Site-directed_mutagenesis site-directed mutagenesis], utilizing suitable expression | [http://en.wikipedia.org/wiki/Molecular_dynamics molecular dynamics] and [http://en.wikipedia.org/wiki/Electrostatics electrostatics] and to [http://en.wikipedia.org/wiki/Site-directed_mutagenesis site-directed mutagenesis], utilizing suitable expression | ||
| - | systems. [http://en.wikipedia.org/wiki/Acetylcholinesterase Acetylcholinesterase] [http://en.wikipedia.org/wiki/Hydrolysis hydrolysizes] the [http://en.wikipedia.org/wiki/Neurotransmitter neurotransmitter] [[acetylcholine]] <scene name='2ace/Ach/4'>(ACh)</scene>, producing <scene name='2ace/Ach/5'>choline and an acetate</scene> group. <scene name='2ace/Ach/4'>ACh</scene> directly binds <scene name='2ace/Ach/11'>Ser200</scene> (via its [http://en.wikipedia.org/wiki/Nucleophile nucleophilic] Oγ atom) within the [http://en.wikipedia.org/wiki/Catalytic_triad catalytic triad] <scene name='2ace/Ach/12'>(Ser200, His440, and Glu327)</scene> (ACh/''Tc''AChE structure [[2ace]]). The residues <scene name='2ace/Ach/13'>Trp84 and Phe330</scene> are also important in the [http://en.wikipedia.org/wiki/Ligand ligand] recognition. See also: [[AChE inhibitors and substrates]] | + | systems. [http://en.wikipedia.org/wiki/Acetylcholinesterase Acetylcholinesterase] [http://en.wikipedia.org/wiki/Hydrolysis hydrolysizes] the [http://en.wikipedia.org/wiki/Neurotransmitter neurotransmitter] [[acetylcholine]] <scene name='2ace/Ach/4'>(ACh)</scene>, producing <scene name='2ace/Ach/5'>choline and an acetate</scene> group. <scene name='2ace/Ach/4'>ACh</scene> directly binds <scene name='2ace/Ach/11'>Ser200</scene> (via its [http://en.wikipedia.org/wiki/Nucleophile nucleophilic] Oγ atom) within the [http://en.wikipedia.org/wiki/Catalytic_triad catalytic triad] <scene name='2ace/Ach/12'>(Ser200, His440, and Glu327)</scene> (ACh/''Tc''AChE structure [[2ace]]). The residues <scene name='2ace/Ach/13'>Trp84 and Phe330</scene> are also important in the [http://en.wikipedia.org/wiki/Ligand ligand] recognition <ref name="Raves">PMID:8989325</ref>. See also: [[AChE inhibitors and substrates]] |
{{Clear}} | {{Clear}} | ||
| Line 30: | Line 30: | ||
====HupA==== | ====HupA==== | ||
| - | '''HupA''', discovered by Chinese scientists from 1980s, has been proved to be a powerful, highly specific, and [http://en.wikipedia.org/wiki/Enzyme_inhibitor#Reversible_inhibitors reversible inhibitor] of AChE. The [http://en.wikipedia.org/wiki/X-ray_crystallography crystal structure] of the complex of ''Tc''AChE with HupA at 2.5 Å resolution ([[1vot]]) was determined in 1997 and it shows an unexpected orientation for the inhibitor with surprisingly few strong direct interactions with protein residues to explain its high affinity. <font color='blueviolet'><b>HupA</b></font> binds to ''Tc''AChE at the active site, and its <scene name='1vot/Active_site/8'>observed orientation is almost orthogonal</scene> in comparison to <font color='gray'><b>ACh</b></font>. The principal interactions of <scene name='1vot/1vot_ache_interactions/2'>HupA with TcAChE</scene> are including: a direct <scene name='1vot/1vot_199_130_117/2'>hydrogen bond with Tyr130 and HBs with Glu199 and Gly117 </scene> <font color='orange'><b>(colored orange)</b></font> through a water molecule as a linker at the bottom of the gorge; [http://en.wikipedia.org/wiki/Cation-pi_interaction cation-π] interactions between the amino group of <scene name='1vot/1vot_84_330/2'>HupA and Trp84 and Phe330</scene> <font color='lime'><b>(colored lime)</b></font> with the distance between the nitrogen and the centroid of the aromatic rings of 4.8 and 4.7 Å, respectively; at the top of the gorge, [http://en.wikipedia.org/wiki/Hydrogen_bond hydrogen bonds] through two water molecules as linkers formed between the amino group of <scene name='1vot/1vot_70_72_81_85_121/3'>HupA and Tyr70, Asp72, Ser81, Asn85 and Tyr121</scene> <font color='magenta'><b>(colored magenta)</b></font>. An unusually short (~3.0 Å) C-H→O HB has been seen between the ethylidene methyl group of <scene name='1vot/1vot_440/2'>HupA and the main chain oxygen of His440</scene> <font color='crimson'><b>(colored crimson)</b></font>. | + | '''HupA''', discovered by Chinese scientists from 1980s, has been proved to be a powerful, highly specific, and [http://en.wikipedia.org/wiki/Enzyme_inhibitor#Reversible_inhibitors reversible inhibitor] of AChE. The [http://en.wikipedia.org/wiki/X-ray_crystallography crystal structure] of the complex of ''Tc''AChE with HupA at 2.5 Å resolution ([[1vot]]) was determined in 1997 and it shows an unexpected orientation for the inhibitor with surprisingly few strong direct interactions with protein residues to explain its high affinity. <font color='blueviolet'><b>HupA</b></font> binds to ''Tc''AChE at the active site, and its <scene name='1vot/Active_site/8'>observed orientation is almost orthogonal</scene> in comparison to <font color='gray'><b>ACh</b></font>. The principal interactions of <scene name='1vot/1vot_ache_interactions/2'>HupA with TcAChE</scene> are including: a direct <scene name='1vot/1vot_199_130_117/2'>hydrogen bond with Tyr130 and HBs with Glu199 and Gly117 </scene> <font color='orange'><b>(colored orange)</b></font> through a water molecule as a linker at the bottom of the gorge; [http://en.wikipedia.org/wiki/Cation-pi_interaction cation-π] interactions between the amino group of <scene name='1vot/1vot_84_330/2'>HupA and Trp84 and Phe330</scene> <font color='lime'><b>(colored lime)</b></font> with the distance between the nitrogen and the centroid of the aromatic rings of 4.8 and 4.7 Å, respectively; at the top of the gorge, [http://en.wikipedia.org/wiki/Hydrogen_bond hydrogen bonds] through two water molecules as linkers formed between the amino group of <scene name='1vot/1vot_70_72_81_85_121/3'>HupA and Tyr70, Asp72, Ser81, Asn85 and Tyr121</scene> <font color='magenta'><b>(colored magenta)</b></font>. An unusually short (~3.0 Å) C-H→O HB has been seen between the ethylidene methyl group of <scene name='1vot/1vot_440/2'>HupA and the main chain oxygen of His440</scene> <font color='crimson'><b>(colored crimson)</b></font> <ref name="Raves"/>. |
====Galanthamine==== | ====Galanthamine==== | ||
| - | <scene name='AChE_inhibitors_and_substrates/Com_view_gal/1'>Galanthamine (GAL, Reminyl)</scene>. <scene name='AChE_inhibitors_and_substrates/Com_view_gal/2'>GAL</scene> <font color='red'><b>(red)</b></font> is an [http://en.wikipedia.org/wiki/Alkaloid alkaloid] from the flower snowdrop ([http://en.wikipedia.org/wiki/Galanthus ''Galanthus nivalis'']). The [http://en.wikipedia.org/wiki/X-ray_crystallography X-ray crystal structure] of the ''Tc''AChE/GAL complex ([[1dx6]]) was determined at 2.3 Å resolution. The inhibitor binds at the base of the [http://en.wikipedia.org/wiki/Active_site active site] gorge of ''Tc''AChE, interacting with both the choline-binding site (Trp84) and the acyl-binding pocket (Phe288, Phe290). The [http://en.wikipedia.org/wiki/Amine tertiary amine] appears to make a non-conventional [http://en.wikipedia.org/wiki/Hydrogen_bond hydrogen bond], via its N-methyl group, to Asp72. The [http://en.wikipedia.org/wiki/Hydroxyl#Hydroxyl_group hydroxyl group] of the inhibitor makes a strong hydrogen bond (2.7 Å) with Glu199. <font color='gray'><b>ACh (gray)</b></font> is shown for comparison. | + | <scene name='AChE_inhibitors_and_substrates/Com_view_gal/1'>Galanthamine (GAL, Reminyl)</scene>. <scene name='AChE_inhibitors_and_substrates/Com_view_gal/2'>GAL</scene> <font color='red'><b>(red)</b></font> is an [http://en.wikipedia.org/wiki/Alkaloid alkaloid] from the flower snowdrop ([http://en.wikipedia.org/wiki/Galanthus ''Galanthus nivalis'']). The [http://en.wikipedia.org/wiki/X-ray_crystallography X-ray crystal structure] of the ''Tc''AChE/GAL complex ([[1dx6]]) was determined at 2.3 Å resolution. The inhibitor binds at the base of the [http://en.wikipedia.org/wiki/Active_site active site] gorge of ''Tc''AChE, interacting with both the choline-binding site (Trp84) and the acyl-binding pocket (Phe288, Phe290). The [http://en.wikipedia.org/wiki/Amine tertiary amine] appears to make a non-conventional [http://en.wikipedia.org/wiki/Hydrogen_bond hydrogen bond], via its N-methyl group, to Asp72. The [http://en.wikipedia.org/wiki/Hydroxyl#Hydroxyl_group hydroxyl group] of the inhibitor makes a strong hydrogen bond (2.7 Å) with Glu199 <ref name="Greenblatt">PMID:10606746</ref>. <font color='gray'><b>ACh (gray)</b></font> is shown for comparison. |
====Tacrine==== | ====Tacrine==== | ||
<scene name='AChE_inhibitors_and_substrates/Com_view_tacrine/1'>Tacrine (Cognex)</scene>. | <scene name='AChE_inhibitors_and_substrates/Com_view_tacrine/1'>Tacrine (Cognex)</scene>. | ||
| - | In the X-ray crystal structure of ''Tc''AChE/<scene name='AChE_inhibitors_and_substrates/Com_view_tacrine/2'>tacrine</scene> complex which was determined at 2.8 Å resolution, the [http://en.wikipedia.org/wiki/Tacrine tacrine] is seen <font color='magenta'><b>(magenta)</b></font> bound in the active site of ''Tc''AChE ([[1acj]]). <font color='gray'><b>ACh (gray)</b></font> is shown for comparison. | + | In the X-ray crystal structure of ''Tc''AChE/<scene name='AChE_inhibitors_and_substrates/Com_view_tacrine/2'>tacrine</scene> complex which was determined at 2.8 Å resolution, the [http://en.wikipedia.org/wiki/Tacrine tacrine] is seen <font color='magenta'><b>(magenta)</b></font> bound in the active site of ''Tc''AChE ([[1acj]]) <ref name="Harel">PMID:8415649</ref>. <font color='gray'><b>ACh (gray)</b></font> is shown for comparison. |
====Rivastigmine==== | ====Rivastigmine==== | ||
| - | <scene name='1gqr/Com_view/1'>Rivastigmine (Exelon)</scene> is a [http://en.wikipedia.org/wiki/Carbamate carbamate] inhibitor of AChE, and it is currenly used in therapy of [http://en.wikipedia.org/wiki/Alzheimer's_disease Alzheimer's disease]. Rivastigmine (colored yellow) interacts with ''Tc''AChE <font color='lime'><b>(colored lime)</b></font> at the <scene name='1gqr/Active_site/4'>active-site gorge</scene> ([[1gqr]]). The carbamyl moiety of rivastigmine is <scene name='1gqr/Active_site/9'>covalently bound</scene> to the active-site S200 Oγ. The second part of rivastigmine (the leaving group), NAP ((−)-S-3-[1-(dimethylamino)ethyl]phenol) is also held in the active-site gorge, but it is <scene name='1gqr/Active_site/6'>separated</scene> from the carbamyl moiety, hence, carbamylation took place. The <scene name='1gqr/Active_site/7'>crystal structure</scene> of ''Tc''AChE/<font color='magenta'><b>NAP (colored magenta)</b></font> is known ([[1gqs]]). The <font color='violet'><b>''Tc''AChE active-site residues</b></font> which are interacting with NAP are <font color='violet'><b>colored violet</b></font>. NAP is located in a similar region of ''Tc''AChE active site, but with different orientation than that of the NAP part (colored yellow) in the ''Tc''AChE/rivastigmine complex. Only H440 and F330 significantly change their side-chain conformations. <scene name='1gqr/Active_site/8'>Overlap</scene> of the ''Tc''AChE active sites in 4 different structures (<font color='lime'><b>''Tc''AChE</b></font>/rivastigmine ([[1gqr]]), <font color='violet'><b>''Tc''AChE</b></font>/<font color='magenta'><b>NAP</b></font> ([[1gqs]]), <font color='cyan'><b>native ''Tc''AChE</b></font> ([[2ace]]), and ''Tc''AChE/'''VX''' ([[1vxr]], ''Tc''AChE colored white and VX black) reveals that the conformation of H440 in the ''Tc''AChE/NAP structure is very similar its conformation in the native ''Tc''AChE ([[2ace]]), but the distance between H440 Nδ and E327 Oε is significantly longer in the ''Tc''AChE/rivastigmine and the ''Tc''AChE/'''VX''' complexes. This structural change disrupts the [http://en.wikipedia.org/wiki/Catalytic_triad catalytic triad] consisting of S200, E327, H440. This could explain the very slow kinetics of AChE reactivation after its inhibition by rivastigmine. | + | <scene name='1gqr/Com_view/1'>Rivastigmine (Exelon)</scene> is a [http://en.wikipedia.org/wiki/Carbamate carbamate] inhibitor of AChE, and it is currenly used in therapy of [http://en.wikipedia.org/wiki/Alzheimer's_disease Alzheimer's disease]. Rivastigmine (colored yellow) interacts with ''Tc''AChE <font color='lime'><b>(colored lime)</b></font> at the <scene name='1gqr/Active_site/4'>active-site gorge</scene> ([[1gqr]]). The carbamyl moiety of rivastigmine is <scene name='1gqr/Active_site/9'>covalently bound</scene> to the active-site S200 Oγ. The second part of rivastigmine (the leaving group), NAP ((−)-S-3-[1-(dimethylamino)ethyl]phenol) is also held in the active-site gorge, but it is <scene name='1gqr/Active_site/6'>separated</scene> from the carbamyl moiety, hence, carbamylation took place. The <scene name='1gqr/Active_site/7'>crystal structure</scene> of ''Tc''AChE/<font color='magenta'><b>NAP (colored magenta)</b></font> is known ([[1gqs]]). The <font color='violet'><b>''Tc''AChE active-site residues</b></font> which are interacting with NAP are <font color='violet'><b>colored violet</b></font>. NAP is located in a similar region of ''Tc''AChE active site, but with different orientation than that of the NAP part (colored yellow) in the ''Tc''AChE/rivastigmine complex. Only H440 and F330 significantly change their side-chain conformations. <scene name='1gqr/Active_site/8'>Overlap</scene> of the ''Tc''AChE active sites in 4 different structures (<font color='lime'><b>''Tc''AChE</b></font>/rivastigmine ([[1gqr]]), <font color='violet'><b>''Tc''AChE</b></font>/<font color='magenta'><b>NAP</b></font> ([[1gqs]]), <font color='cyan'><b>native ''Tc''AChE</b></font> ([[2ace]]), and ''Tc''AChE/'''VX''' ([[1vxr]], ''Tc''AChE colored white and VX black) reveals that the conformation of H440 in the ''Tc''AChE/NAP structure is very similar its conformation in the native ''Tc''AChE ([[2ace]]), but the distance between H440 Nδ and E327 Oε is significantly longer in the ''Tc''AChE/rivastigmine and the ''Tc''AChE/'''VX''' complexes. This structural change disrupts the [http://en.wikipedia.org/wiki/Catalytic_triad catalytic triad] consisting of S200, E327, H440. This could explain the very slow kinetics of AChE reactivation after its inhibition by rivastigmine <ref name="Bar-On">PMID:11888271</ref>. |
=== The second generation of AD drugs - bivalent AChE inhibitors === | === The second generation of AD drugs - bivalent AChE inhibitors === | ||
Revision as of 07:54, 28 November 2010
|
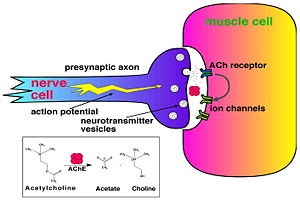
Acetylcholinesterase (AChE) is key enzyme in the nervous system of animals. By rapid hydrolysis of the neurotransmitter, acetylcholine (ACh), AChE terminates neurotransmission at cholinergic synapses. It is a very fast enzyme, especially for a serine hydrolase, functioning at a rate approaching that of a diffusion-controlled reaction. AChE inhibitors are among the key drugs approved by the FDA for management of Alzheimer's disease (AD). The powerful toxicity of organophosphorus (OP) poisons is attributed primarily to their potent AChE inhibitors.
Key Enzyme in the Nervous System
|
Solution of the three-dimensional (3D) structure of Torpedo californica acetylcholinesterase (TcAChE) in 1991 opened up new horizons in research on an enzyme that had already been the subject of intensive investigation.[1] The unanticipated structure of this extremely rapid enzyme, in which the active site was found to be buried at the bottom of a , lined by (colored dark magenta), led to a revision of the views then held concerning substrate traffic, recognition and hydrolysis.[2] To understand how those aromatic residues behave with the enzyme, see Flexibility of aromatic residues in acetylcholinesterase. Solution of the 3D structure of acetylcholinesterase led to a series of theoretical and experimental studies, which took advantage of recent advances in theoretical techniques for treatment of proteins, such as molecular dynamics and electrostatics and to site-directed mutagenesis, utilizing suitable expression systems. Acetylcholinesterase hydrolysizes the neurotransmitter acetylcholine , producing group. directly binds (via its nucleophilic Oγ atom) within the catalytic triad (ACh/TcAChE structure 2ace). The residues are also important in the ligand recognition [3]. See also: AChE inhibitors and substrates
Treatment of Alzheimer's disease
Alzheimer's disease (AD) is a disorder that attacks the central nervous system through progressive degeneration of its neurons. AD occurs in around 10% of the elderly and, as yet, there is no known cure. Patients with this disease develop dementia which becomes more severe as the disease progresses. It was suggested that symptoms of AD are caused by decrease of activity of cholinergic neocortical and hippocampal neurons. Treatment of AD by ACh precursors and cholinergic agonists was ineffective or caused severe side effects. ACh hydrolysis by AChE causes termination of cholinergic neurotransmission. Therefore, compounds which inhibit AChE might significantly increase the levels of ACh depleted in AD. Indeed, it was shown that AChE inhibitors improve the cognitive abilities of AD patients at early stages of the disease development.
| |||||||||||
Organophosphorus acid anhydride nerve agents
|
Organophosphorus (OP) acid anhydride nerve agents are potent inhibitors which rapidly phosphonylate AChE and then may undergo an internal dealkylation reaction (called "aging") to produce an OP-enzyme conjugate that cannot be reactivated. As was mentioned above, AChE hydrolysizes the neurotransmitter , producing group. directly binds catalytic (via its nucleophilic Oγ atom). , O-(1,2,2-trimethylpropyl) methylphosphonofluoridate (fluorine atom is colored violet and phosphorus atom is colored darkmagenta), is one of the most toxic OPs. Soman inhibits AChE by to catalytic Ser200, . This process implicates nucleophilic attack of the Ser200 nucleophilic Oγ atom on the phosphorus atom of soman, with concomitant departure of its fluoride atom. After that AChE catalyzes the of the soman or other OP. This causes irreversible inhibition of AChE, "aged" soman/AChE conjugate can not be reactivated. However, before “aging”, at the step of , AChE can be by nucleophiles, such as pralidoxime (2-PAM), resulting in of the phosphonyl adduct from Ser200 Oγ. At the (2wfz) the catalytic His440 forms hydrogen bonds with Ser200 Oγ and Glu327 Oε1 via its Nε2 and Nδ1 nitrogens, respectively. The O2 atom of soman is within hydrogen bonding distance of His440 Nε2. Soman O1 mimicks carbonyl oxygen of ACh. A water molecule 1001 interacting with soman O2 is represented as a red ball. The active site residues of the nonaged soman/TcAChE are colored yellow. The O2 atom of the (2wg0) forms a salt bridge with His440 Nε2. The active site residues of the aged soman/TcAChE are colored pink. of the structures of the nonaged (2wfz) and aged (2wg0) conjugates reveals a small, but important, change within the active site - the imidazole ring of His440 is tilted back to a native-like conformation after dealkylation. The water molecule 1001, which interacts with soman O2 in the nonaged crystal structure, is not within hydrogen bonding distance of O2 in the aged crystal structure. 2-PAM binds poorly to the nonaged phosphonylated enzyme (its electron density was not found) and binds in an after soman aging to TcAChE (2wg1).
Selected 3D Structures of AChE
Acetylcholinesterase - AChE native
3lii – hAChE - recombinant human
1ea5, 2ace – TcAChE – trigonal – Torpedo californica
2j3d – TcAChE – monoclinic
1w75 – TcAChE – orthorhombic
1eea – TcAChE – cubic
2vt6, 2vt7 – TcAChE – different dosage
1qid to 1qim - TcAChE synchrotron radiation damage
1j06, 1maa – mAChE - mouse
1qo9 – DmAChE - Drosophila
1c2o, 1c2b – electrophorus AChE – Electric eel
AChE inhibitors (In Different Languages)
1eve AChE-Aricept complex, 1eve (Arabic), 1eve (Chinese), 1eve (Italian), 1eve (Russian), 1eve (Spanish), 1eve (Turkish)
1vot AChE-Huperzine A complex, 1vot (Chinese)
AChE active site inhibitors conjugating at the bottom of the active site gorge
2w9i – TcAChE + methylene blue
2wls – MosAChE + AMTS13
2vq6 – TcAChE + 2-PAM
2j3q – TcAChE + Thioflavin T
2ha0 – mAChE + ketoamyltrimethylammonium
2h9y – mAChE + TMTFA
1gpk, 1gpn, 1vot – TcAChE + huperzine
1gqr – TcAChE + rivastigmine
1gqs – TcAChE + NAP
1e66 – TcAChE + huprine
1dx4, 1qon – DmAChE + tacrine derivative
1oce – TcAChE + MF268
1ax9, 1ack – TcAChE + edrophonium
1amn – TcAChE + TMTFA
1acj – TcAChE + tacrine
AChE peripheral site inhibitors conjugating at the surface of the protein
1ku6 - mAChE + fasciculin 2
1ku6, 1mah - mAChE + fasciculin 2
1j07 - mAChE + decidium
1n5m - mAChE + gallamine
1n5r - mAChE + propidium
1b41, 1f8u - hAChE + fasciculin 2
1fss - TcAChE + fasciculin 2
AChE bis inhibitors spanning the active site gorge
3i6m – TcAChE + N-piperidinopropyl galanthamine
3i6z - TcAChE + saccharinohexyl galanthamine
1zgb, 1zgc – TcAChE + tacrine (10) hupyridone
2w6c – TcAChE + bis-(-)-nor-meptazinol
2ckm, 2cmf – TcAChE + bis-tacrine
2cek – TcAChE + N-[8-(1,2,3,4-tetrahydroacridin-9-ylthio)octyl]-1,2,3,4-tetrahydroacridin-9-amine
1ut6 - TcAChE + N-9-(1,2,3,4-tetrahydroacridinyl)-1,8-diaminooctane
1odc - TcAChE + N-4-quinolyl-N-9-(1,2,3,4-tetrahydroacridinyl)-1,8-diaminooctane
1w4l, 1w6r, 1w76, 1dx6, 1qti - TcAChE + galanthamine and derivative
1q83, 1q84 - mAChE + TZ2PA6
1h22, 1h23 – TcAChE + bis-hupyridone
1hbj – TcAChE + quinoline derivativev
1e3q – TcAChE + bw284c51
1eve – TcAChE + e2020
1acl – TcAChE + decamethonium
AChE organophosphate inhibitors causing irreversible inhibition
2wu3 – mAChE + fenamiphos and HI-6
2wu4 – mAChE + fenamiphos and ortho-7
2jgf - mAChE + fenamiphos
2wfz, 2wg0, 1som - TcAChE + soman
2wg1 - TcAChE + soman + 2-PAM
2whp, 2whq, 2whr – mAChE + sarin and HI-6
2jgg - mAChE + sarin
2jgl - mAChE + VX and sarin
1cfj - TcAChE + sarin, GB
3dl4, 3dl7 – mAChE + tabun
2jey – mAChE + HLO-7
2c0p, 2c0q - mAChE + tabun
2jez - mAChE + tabun + HLO-7
2jf0 - mAChE + tabun + Ortho-7
2jgh - mAChE + VX
1vxo, 1vxr - TcAChE + VX
2jgi, 2jgm - mAChE + DFP
1dfp - TcAChE + DFP
2jgj, 2jgk, 2jge - mAChE + methamidophos
2gyu - mAChE + HI-6
2gyv - mAChE + Ortho-7
2gyw - mAChE + obidoxime
AChE substrate analogues mimicking the binding of the substrate acetylcholine
2ha4 – mAChE (mutant) + acetylcholine
2vja, 2vjb, 2vjc, 2vjd, 2cf5 – TcAChE + 4-oxo-N,N,N-trimethylpentanaminium
2v96, 2v97, 2v98, 2v99 – TcAChE + 1-(2-nitrophenyl)-2,2,2-trifluoroethyl-arsenocholine
2ha2 – mAChE + succinylcholine
2ha3 - mAChE + choline
2ha5 – mAChE (mutant) + acetylthiocholine
2ha6 – mAChE (mutant) + succinylthiocholine
2ha7 – mAChE (mutant) + butyrylthiocholine
2ch4, 2c58 – TcAChE + acetylthiocholine
2c5g – TcAChE + thiocholine
Others...
2j4f – TcAChE + Hg
1vzj – TcAChE tetramerization domain
1jjb – TcAChE + PEG
Additional Resources
For additional information, see:
Alzheimer's Disease
AChE inhibitors and substrates
AChE inhibitors and substrates (Part II)
AChE inhibitors and substrates (Part III)
AChE bivalent inhibitors
AChE bivalent inhibitors (Part II)
External Links
- Acetylcholinesterase Tutorial by Karl Oberholser, Messiah College
- PDB Molecule of the Month - Acetylcholinesterase
- Movies: X-ray Damage in ACh & Nature's Vacuum Cleaner by R. Gillilan, Cornell Univ
References
- ↑ Sussman JL, Harel M, Frolow F, Oefner C, Goldman A, Toker L, Silman I. Atomic structure of acetylcholinesterase from Torpedo californica: a prototypic acetylcholine-binding protein. Science. 1991 Aug 23;253(5022):872-9. PMID:1678899
- ↑ Botti SA, Felder CE, Lifson S, Sussman JL, Silman I. A modular treatment of molecular traffic through the active site of cholinesterase. Biophys J. 1999 Nov;77(5):2430-50. PMID:10545346
- ↑ 3.0 3.1 Raves ML, Harel M, Pang YP, Silman I, Kozikowski AP, Sussman JL. Structure of acetylcholinesterase complexed with the nootropic alkaloid, (-)-huperzine A. Nat Struct Biol. 1997 Jan;4(1):57-63. PMID:8989325
- ↑ Greenblatt HM, Kryger G, Lewis T, Silman I, Sussman JL. Structure of acetylcholinesterase complexed with (-)-galanthamine at 2.3 A resolution. FEBS Lett. 1999 Dec 17;463(3):321-6. PMID:10606746
- ↑ Harel M, Schalk I, Ehret-Sabatier L, Bouet F, Goeldner M, Hirth C, Axelsen PH, Silman I, Sussman JL. Quaternary ligand binding to aromatic residues in the active-site gorge of acetylcholinesterase. Proc Natl Acad Sci U S A. 1993 Oct 1;90(19):9031-5. PMID:8415649
- ↑ Bar-On P, Millard CB, Harel M, Dvir H, Enz A, Sussman JL, Silman I. Kinetic and structural studies on the interaction of cholinesterases with the anti-Alzheimer drug rivastigmine. Biochemistry. 2002 Mar 19;41(11):3555-64. PMID:11888271
Proteopedia Page Contributors and Editors (what is this?)
Michal Harel, Joel L. Sussman, Alexander Berchansky, David Canner, Eran Hodis, Clifford Felder, Jaime Prilusky, Harry Greenblatt, Yechun Xu