This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Magainin 2
From Proteopedia
| Line 3: | Line 3: | ||
==Introduction== | ==Introduction== | ||
| - | Magainin are a class of antimicrobial peptides (AMPs) found in the African clawed frog Xenopus Laevis. | + | '''Magainin''' are a class of antimicrobial peptides (AMPs) found in the African clawed frog Xenopus Laevis. |
AMPs consists of 10-50 amino acids, and are produced by Eukaryotes, as part of their defence mechanism from bacteria. For informatoin about AMPs you can visit the Proteopedia page [[Antimicrobial peptides]] | AMPs consists of 10-50 amino acids, and are produced by Eukaryotes, as part of their defence mechanism from bacteria. For informatoin about AMPs you can visit the Proteopedia page [[Antimicrobial peptides]] | ||
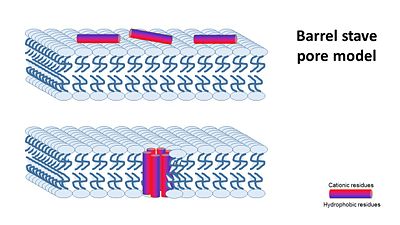
Magainin 1 and 2 were discovered by Dr. Michael Zasloff and first reported in 1987. They have an alpha helix structure, and are water soluble and Potentially amphiphilic. | Magainin 1 and 2 were discovered by Dr. Michael Zasloff and first reported in 1987. They have an alpha helix structure, and are water soluble and Potentially amphiphilic. | ||
| Line 57: | Line 57: | ||
</StructureSection> | </StructureSection> | ||
| + | |||
| + | ==3D structures of magainin 2== | ||
| + | Updated on {{REVISIONDAY2}}-{{MONTHNAME|{{REVISIONMONTH}}}}-{{REVISIONYEAR}} | ||
| + | |||
| + | [[2mag]], [[2lsa]] – Mag2 – frog - NMR<br /> | ||
| + | [[1dum]] – Mag2 (mutant) - NMR<br /> | ||
| + | [[4mgp]], [[5cgn]], [[5cgo]] – Mag2 (mutant) <br /> | ||
| + | [[1d9j]], [[1d9l]], [[1d9o]], [[1d9m]], [[1d9p]], [[1f0d]], [[1f0e]], [[1f0f]], [[1f0g]], [[1f0h]] – Mag2/cecropin A - NMR<br /> | ||
| + | |||
== References == | == References == | ||
1-'''J. Gesella., M. Zasloffb and S. J. Opellaa'''., Two-dimensional H NMR experiments show that the 23-residue magainin antibiotic peptide is an α-helix in dodecylphosphocholine micelles, sodium dodecylsulfate micelles, and trifluoroethanol/water solution. ''Journal of Biomolecular NMR'', 1997. 9: 127–135. | 1-'''J. Gesella., M. Zasloffb and S. J. Opellaa'''., Two-dimensional H NMR experiments show that the 23-residue magainin antibiotic peptide is an α-helix in dodecylphosphocholine micelles, sodium dodecylsulfate micelles, and trifluoroethanol/water solution. ''Journal of Biomolecular NMR'', 1997. 9: 127–135. | ||
Revision as of 13:15, 6 January 2016
Magainin 2
| |||||||||||
3D structures of magainin 2
Updated on 06-January-2016
2mag, 2lsa – Mag2 – frog - NMR
1dum – Mag2 (mutant) - NMR
4mgp, 5cgn, 5cgo – Mag2 (mutant)
1d9j, 1d9l, 1d9o, 1d9m, 1d9p, 1f0d, 1f0e, 1f0f, 1f0g, 1f0h – Mag2/cecropin A - NMR
References
1-J. Gesella., M. Zasloffb and S. J. Opellaa., Two-dimensional H NMR experiments show that the 23-residue magainin antibiotic peptide is an α-helix in dodecylphosphocholine micelles, sodium dodecylsulfate micelles, and trifluoroethanol/water solution. Journal of Biomolecular NMR, 1997. 9: 127–135.
2-Z. Hayouka., D. E. Mortenson., D. F. Kreitler., B. Weisblum., K. T. Forest, and S. H. Gellman., Evidence for Phenylalanine Zipper-Mediated Dimerization in the X‑ray Crystal Structure of a Magainin 2 Analogue. J. Am. Chem. Soc, 2013. 135: 15738−15741.
3- Y. Tamba & M. Yamazaki.,Magainin 2-Induced Pore Formation in the Lipid Membranes Depends on Its Concentration in the Membrane Interface. J. Phys. Chem. B, 2009. 113: 4846–4852.
4- W. C. Wimley., Describing the Mechanism of Antimicrobial Peptide Action with the Interfacial Activity Model. ACS CHEMICAL BIOLOGY, 2010. 10: 905-917.