This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Acyl carrier protein synthase
From Proteopedia
| Line 5: | Line 5: | ||
{{Clear}} | {{Clear}} | ||
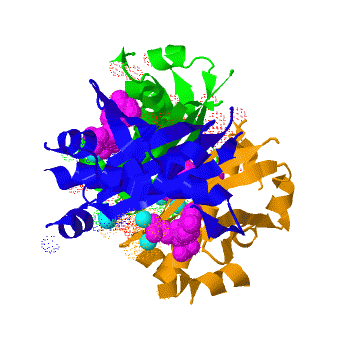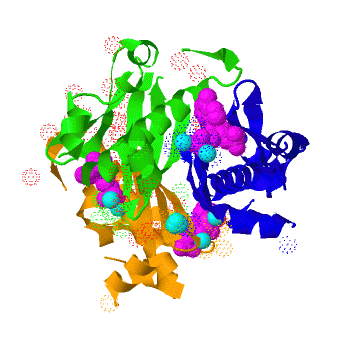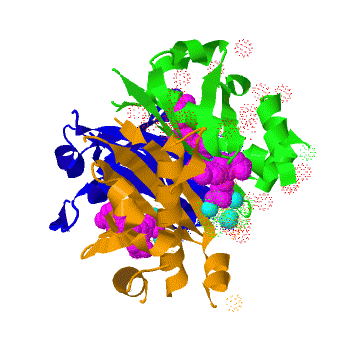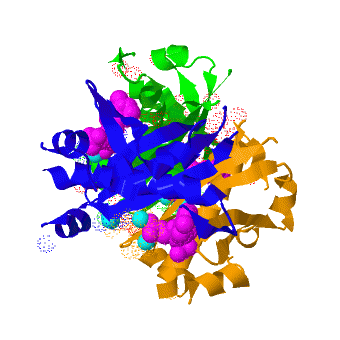
| - | <scene name='3hqj/Align/2'>Structural alignment</scene> of the structures of the ''Mtb'' AcpS trimer (in <span style="color:lime;background-color:black;font-weight:bold;">green</span>, <font color='blue'><b>blue</b></font>, and <span style="color:orange;background-color:black;font-weight:bold;">orange</span>) and the ''B. subtilis'' AcpS trimer ([[1f7t]], in <font color='red'><b>red</b></font>, <span style="color:cyan;background-color:black;font-weight:bold;">cyan</span>, and < | + | <scene name='3hqj/Align/2'>Structural alignment</scene> of the structures of the ''Mtb'' AcpS trimer (in <span style="color:lime;background-color:black;font-weight:bold;">green</span>, <font color='blue'><b>blue</b></font>, and <span style="color:orange;background-color:black;font-weight:bold;">orange</span>) and the ''B. subtilis'' AcpS trimer ([[1f7t]], in <font color='red'><b>red</b></font>, <span style="color:cyan;background-color:black;font-weight:bold;">cyan</span>, and <span style="color:yellow;background-color:black;font-weight:bold;">yellow</span>) reveals that the ''Mtb'' AcpS structure is similar to those of other members of group I phosphopantetheine transferase (PPT) family. The <scene name='3hqj/Align/3'>important difference</scene> is that the extended α3 helix of ''Mtb'' AcpS has open conformation. Such open conformation permits to the extended loop of one monomer (<span style="color:lime;background-color:black;font-weight:bold;">green</span>) to interact with adjacent monomer (<font color='blue'><b>blue</b></font>). The considerably shorter α3 of one ''B. subtilis'' AcpS monomer (<font color='red'><b>red</b></font>) has closed conformation and this doesn't allow interaction with the neighboring monomer (<span style="color:cyan;background-color:black;font-weight:bold;">cyan</span>). |
{{Clear}} | {{Clear}} | ||
Revision as of 11:55, 17 May 2015
| |||||||||||
3D structures of acyl carrier protein synthase
Updated on 17-May-2015
References
- Parris KD, Lin L, Tam A, Mathew R, Hixon J, Stahl M, Fritz CC, Seehra J, Somers WS. Crystal structures of substrate binding to Bacillus subtilis holo-(acyl carrier protein) synthase reveal a novel trimeric arrangement of molecules resulting in three active sites. Structure. 2000 Aug 15;8(8):883-95. PMID:10997907
- Dym O, Albeck S, Peleg Y, Schwarz A, Shakked Z, Burstein Y, Zimhony O. Structure-function analysis of the acyl carrier protein synthase (AcpS) from Mycobacterium tuberculosis. J Mol Biol. 2009 Nov 6;393(4):937-50. Epub 2009 Sep 3. PMID:19733180 doi:10.1016/j.jmb.2009.08.065
Proteopedia Page Contributors and Editors (what is this?)
Categories: Topic Page | Mycobacterium tuberculosis | Albeck,S. | Burstein,Y. | Dym,O. | ISPC, Israel Structural Proteomics Center. | Peleg,Y. | Schwarz,A. | Shakked,Z. | Zimhony,O. | An/fold | Cytoplasm | Fatty acid biosynthesis | ISPC | Israel Structural Proteomics Center | Lipid synthesis | Magnesium | Metal-binding | Structural genomic | Transferase