This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Chaperonin
From Proteopedia
(Difference between revisions)
| Line 1: | Line 1: | ||
[[Image:1pcq.png|left|200px|thumb|Crystal Structure of Chaperonin, [[1pcq]]]] | [[Image:1pcq.png|left|200px|thumb|Crystal Structure of Chaperonin, [[1pcq]]]] | ||
| - | {{STRUCTURE_1pcq| PDB=1pcq | SIZE=400| SCENE=Chaperonin/Groel_groes_comnplex/1 |right|CAPTION=GroEL/GroES complex with ADP, AlF3, Mg+2 and K+ ions, [[1pcq]] }} | ||
| - | <StructureSection load='1pcq' size='350' side='right' caption='GroEL/GroES complex with ADP, AlF3, Mg+2 and K+ ions (PDB entry [[1pcq]])' scene=''> | + | <StructureSection load='1pcq' size='350' side='right' caption='GroEL/GroES complex with ADP, AlF3, Mg+2 and K+ ions (PDB entry [[1pcq]])' scene='Chaperonin/Groel_groes_comnplex/1'> |
'''Chaperonins''' (CPN) are oligomeric proteins that mediate the folding of polypeptide chains. Group I CPN are found in bacteria, chloroplasts and mitochondria. For an introductory overview, see [http://en.wikipedia.org/wiki/Chaperonins Chaperonins in Wikipedia]. | '''Chaperonins''' (CPN) are oligomeric proteins that mediate the folding of polypeptide chains. Group I CPN are found in bacteria, chloroplasts and mitochondria. For an introductory overview, see [http://en.wikipedia.org/wiki/Chaperonins Chaperonins in Wikipedia]. | ||
Revision as of 11:30, 6 December 2015
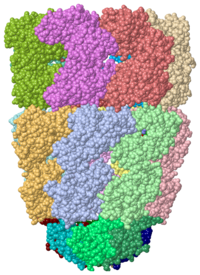
Crystal Structure of Chaperonin, 1pcq
| |||||||||||
3D Structures of Chaperonin
Updated on 06-December-2015
References
Proteopedia Page Contributors and Editors (what is this?)
Michal Harel, Alexander Berchansky, Joel L. Sussman, Eric Martz, Jaime Prilusky
