This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Aminotransferase
From Proteopedia
(Difference between revisions)
| Line 27: | Line 27: | ||
== Structural highlights == | == Structural highlights == | ||
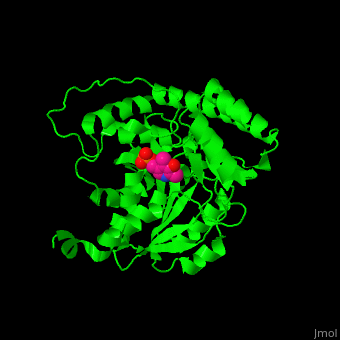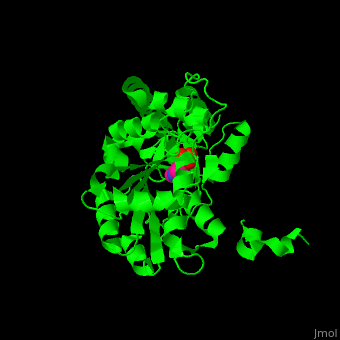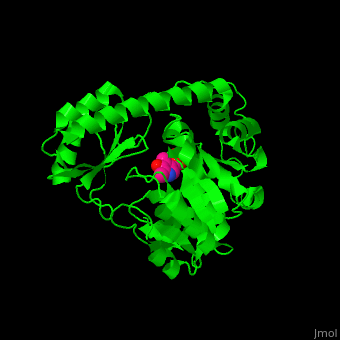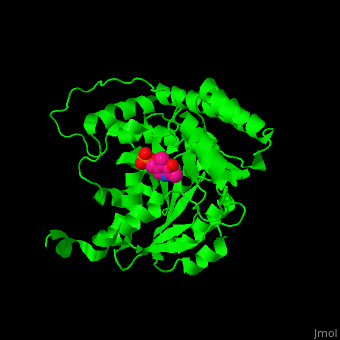
| - | The active site of histidinol-phosphate aminotransferase contains PLP.<ref>PMID:11294630</ref> | + | The <scene name='72/721044/Cv/2'>active site of histidinol-phosphate aminotransferase contains PLP</scene>.<ref>PMID:11294630</ref> |
Revision as of 12:44, 30 December 2015
| |||||||||||
3D structures of aminotransferase
Updated on 30-December-2015
References
- ↑ Mizuguchi H, Hayashi H, Miyahara I, Hirotsu K, Kagamiyama H. Characterization of histidinol phosphate aminotransferase from Escherichia coli. Biochim Biophys Acta. 2003 Apr 11;1647(1-2):321-4. PMID:12686152
- ↑ Kirsch JF, Eichele G, Ford GC, Vincent MG, Jansonius JN, Gehring H, Christen P. Mechanism of action of aspartate aminotransferase proposed on the basis of its spatial structure. J Mol Biol. 1984 Apr 15;174(3):497-525. PMID:6143829 doi:http://dx.doi.org/10.1016/0022-2836(84)90333-4
- ↑ Kirsch JF, Eichele G, Ford GC, Vincent MG, Jansonius JN, Gehring H, Christen P. Mechanism of action of aspartate aminotransferase proposed on the basis of its spatial structure. J Mol Biol. 1984 Apr 15;174(3):497-525. PMID:6143829 doi:http://dx.doi.org/10.1016/0022-2836(84)90333-4
- ↑ Haruyama K, Nakai T, Miyahara I, Hirotsu K, Mizuguchi H, Hayashi H, Kagamiyama H. Structures of Escherichia coli histidinol-phosphate aminotransferase and its complexes with histidinol-phosphate and N-(5'-phosphopyridoxyl)-L-glutamate: double substrate recognition of the enzyme. Biochemistry. 2001 Apr 17;40(15):4633-44. PMID:11294630
Proteopedia Page Contributors and Editors (what is this?)
Michal Harel, Alexander Berchansky, Joel L. Sussman, Jaime Prilusky