This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Sandbox Reserved 1123
From Proteopedia
| Line 1: | Line 1: | ||
| - | === Hello === | ||
| - | |||
<Structure load='3H47' size='350' frame='true' align='right' caption='Structure of Human Immunodeficiency Virus hexameric capsid' scene='' /> | <Structure load='3H47' size='350' frame='true' align='right' caption='Structure of Human Immunodeficiency Virus hexameric capsid' scene='' /> | ||
| Line 31: | Line 29: | ||
== Capsid as therapeutical target == | == Capsid as therapeutical target == | ||
| - | thank to its central role in viral infectious process (genome protection,enveloppe cohesion,uncoating), HIV capsid is a very godd target for antiviral drugs. As a result, this | + | thank to its central role in viral infectious process (genome protection,enveloppe cohesion,uncoating), HIV capsid is a very godd target for antiviral drugs. As a result, many researchs teams are working in this way and some molecules such as ............... are very promising. |
== References == | == References == | ||
Revision as of 12:44, 26 January 2016
|
Contents |
Introduction
Function
Structural highlights
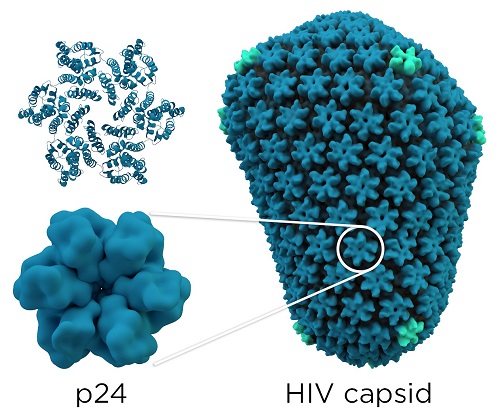
As you can see on the figure bellow, each monomer of capsid is linked to five others to form a hexamer. These hexamers (approximately 330 per virus) associates themselves to form a non-symetrical protein complex.
Interactions with others partners
Even if p24 is classified as a structural protein, it is also involved in many cellular infection processes.
You can find bellow in a non exhaustive list of p24 partners :
- Cytoskeleton (MAP1A, MAP1S, CKAP1, WIRE) - Cyclophylin A - Nuclear pore (certainly but not sure) - Dynein
The HIV-1 capsid acts like a kind of "nuclear localisation signal" because it targets directly the virus toward the nucleus, where the integation takes place.
Capsid as therapeutical target
thank to its central role in viral infectious process (genome protection,enveloppe cohesion,uncoating), HIV capsid is a very godd target for antiviral drugs. As a result, many researchs teams are working in this way and some molecules such as ............... are very promising.
References
Structural image : By Thomas Splettstoesser (www.scistyle.com) (Own work) [CC BY-SA 4.0 (http://creativecommons.org/licenses/by-sa/4.0)], via Wikimedia Commons
Cytoskeleton interactions :
- Fernandez J, Gärtner K, Becker A, et al. HIV-1 capsid interacts with cytoskeletal-associated proteins for intracytoplasmic routing to the nucleus. Retrovirology. 2013;10(Suppl 1):P34. doi:10.1186/1742-4690-10-S1-P34.
- Fernandez J, Portilho DM, Danckaert A, Munier S, Becker A, Roux P, Zambo A, Shorte S, Jacob Y, Vidalain PO, Charneau P, Clavel F, Arhel NJ. Microtubule-associated proteins 1 (MAP1) promote human immunodeficiency virus type I (HIV-1) intracytoplasmic routing to the nucleus. J Biol Chem. 2015 Feb 20;290(8):4631-46. doi: 10.1074/jbc.M114.613133. Epub 2014 Dec 11.
Cyclophylin A interaction :
- Marisa S. Briones, Charles W. Dobard and Samson A. Chow. Role of Human Immunodeficiency Virus Type 1 Integrase in Uncoating of the Viral Core. Accepted manuscript posted online 10 March 2010, doi:.1128/JVI.02382-09 J. Virol. May 2010 vol. 84 no. 10 5181-5190
Nuclear import :
HIV-1 capsid: the multifaceted key player in HIV-1 infection Nature Reviews Microbiology 13,471–483 (2015)doi:10.1038/nrmicro3503