This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
User:Carlos Henrique Passos/Sandbox1
From Proteopedia
(Difference between revisions)
| Line 32: | Line 32: | ||
| - | '''Active Site and Substrate Recognition | + | ===Bonding=== |
| + | |||
| + | '''Disulfide Bridges''' | ||
| + | |||
| + | Although <scene name='78/786632/Proteinase_k/1'>Proteinase K</scene> presents five cysteine amino acids, only four of them participates in disulfide bridges. | ||
| + | There are show two <scene name='78/786632/Disulfide_bridges/1'>disulfide bridges</scene> on this protein. These bridges are made by the presence of cysteines on the central beta-sheet, on the positions 34 and 178 (Cys34 and Cys178). One disulfide bridge is the <scene name='78/786632/Cys34_cys123_bridge/1'>Cys34-Cys123</scene> interactions, and the other is <scene name='78/786632/Cys178_cys249_bridge/1'>Cys178-Cys249</scene> <ref>Betzel, Christian, Gour P. PAL, and Wolfram SAENGER. "Three‐dimensional structure of proteinase K at 0.15‐nm resolution." The FEBS Journal 178.1 (1988): 155-171. Available: https://onlinelibrary.wiley.com/doi/abs/10.1111/j.1432-1033.1988.tb14440.x</ref>. The fifth <scene name='78/786632/Free_cys_73/1'>Cysteine 73</scene> is not participating in any disulfide bridge. | ||
| + | |||
| + | ---- | ||
| + | ---- | ||
| + | |||
| + | '''Hidrogen Bonds''' | ||
| + | |||
| + | The participating amino acids in hydrogen bonding between peptides include 182 out of the total 279 (or 65.2%) <ref>Betzel, Christian, Gour P. PAL, and Wolfram SAENGER. "Three‐dimensional structure of proteinase K at 0.15‐nm resolution." The FEBS Journal 178.1 (1988): 155-171. Available: https://onlinelibrary.wiley.com/doi/abs/10.1111/j.1432-1033.1988.tb14440.x</ref>. | ||
| + | |||
| + | The folding of the protein is stabilized by a total of 130 hydrogen bonds, of which 55 are made between the main chain and side chain functional groups, and 75 are made between side chains. If we include the water molecules, the hydrogen bonds interactions rise up to 191 interactions. <ref>Betzel, Christian, Gour P. PAL, and Wolfram SAENGER. "Three‐dimensional structure of proteinase K at 0.15‐nm resolution." The FEBS Journal 178.1 (1988): 155-171. Available: https://onlinelibrary.wiley.com/doi/abs/10.1111/j.1432-1033.1988.tb14440.x</ref>. | ||
| + | |||
| + | ===Active Site and Substrate Recognition=== | ||
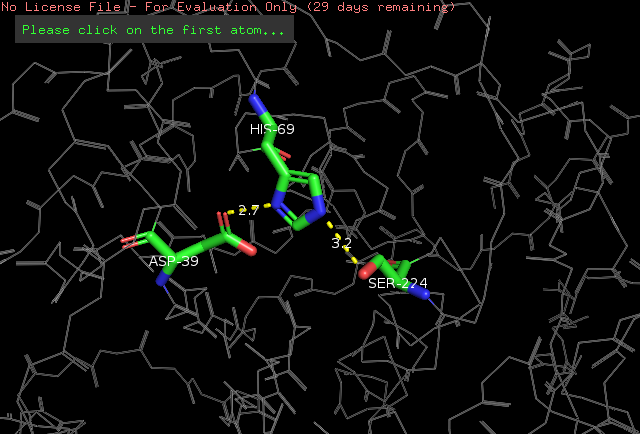
The <scene name='78/786632/Active_site/3'>catalytic site</scene> of Proteinase K is made by a triade of aminoacids, <scene name='78/786632/Asp39/2'>Asp39</scene>, <scene name='78/786632/His69/2'>His69</scene> and <scene name='78/786632/Ser224/2'>Ser224</scene> <ref>Betzel, Christian, Gour P. PAL, and Wolfram SAENGER. "Three‐dimensional structure of proteinase K at 0.15‐nm resolution." The FEBS Journal 178.1 (1988): 155-171. Available: https://onlinelibrary.wiley.com/doi/abs/10.1111/j.1432-1033.1988.tb14440.x</ref>. The pink color indicates that these aminoacids are all polars. | The <scene name='78/786632/Active_site/3'>catalytic site</scene> of Proteinase K is made by a triade of aminoacids, <scene name='78/786632/Asp39/2'>Asp39</scene>, <scene name='78/786632/His69/2'>His69</scene> and <scene name='78/786632/Ser224/2'>Ser224</scene> <ref>Betzel, Christian, Gour P. PAL, and Wolfram SAENGER. "Three‐dimensional structure of proteinase K at 0.15‐nm resolution." The FEBS Journal 178.1 (1988): 155-171. Available: https://onlinelibrary.wiley.com/doi/abs/10.1111/j.1432-1033.1988.tb14440.x</ref>. The pink color indicates that these aminoacids are all polars. | ||
| Line 47: | Line 63: | ||
The <scene name='78/786632/Substrate_recognition/1'>susbtrate recognition</scene> are made by two peptide chains, one that englobes aminoacids ranging from 99 to 104 positions, and the other made of aminoacids ranging from 132 to 136 positions, which can be viewed <scene name='78/786632/Substrate_recognition/2'>here</scene>. The colors indicate where there is a region with alpha-helix (in pink), beta-sheet (in gold), or loop (in white). | The <scene name='78/786632/Substrate_recognition/1'>susbtrate recognition</scene> are made by two peptide chains, one that englobes aminoacids ranging from 99 to 104 positions, and the other made of aminoacids ranging from 132 to 136 positions, which can be viewed <scene name='78/786632/Substrate_recognition/2'>here</scene>. The colors indicate where there is a region with alpha-helix (in pink), beta-sheet (in gold), or loop (in white). | ||
| - | + | ===Ligands=== | |
| - | + | ||
| - | '''Disulfide Bridges''' | ||
| - | |||
| - | Although <scene name='78/786632/Proteinase_k/1'>Proteinase K</scene> presents five cysteine amino acids, only four of them participates in disulfide bridges. | ||
| - | There are show two <scene name='78/786632/Disulfide_bridges/1'>disulfide bridges</scene> on this protein. These bridges are made by the presence of cysteines on the central beta-sheet, on the positions 34 and 178 (Cys34 and Cys178). One disulfide bridge is the <scene name='78/786632/Cys34_cys123_bridge/1'>Cys34-Cys123</scene> interactions, and the other is <scene name='78/786632/Cys178_cys249_bridge/1'>Cys178-Cys249</scene> <ref>Betzel, Christian, Gour P. PAL, and Wolfram SAENGER. "Three‐dimensional structure of proteinase K at 0.15‐nm resolution." The FEBS Journal 178.1 (1988): 155-171. Available: https://onlinelibrary.wiley.com/doi/abs/10.1111/j.1432-1033.1988.tb14440.x</ref>. The fifth <scene name='78/786632/Free_cys_73/1'>Cysteine 73</scene> is not participating in any disulfide bridge. | ||
| - | |||
| - | ---- | ||
| - | ---- | ||
| - | |||
| - | '''Hidrogen Bonds''' | ||
| - | |||
| - | The participating amino acids in hydrogen bonding between peptides include 182 out of the total 279 (or 65.2%) <ref>Betzel, Christian, Gour P. PAL, and Wolfram SAENGER. "Three‐dimensional structure of proteinase K at 0.15‐nm resolution." The FEBS Journal 178.1 (1988): 155-171. Available: https://onlinelibrary.wiley.com/doi/abs/10.1111/j.1432-1033.1988.tb14440.x</ref>. | ||
| - | |||
| - | The folding of the protein is stabilized by a total of 130 hydrogen bonds, of which 55 are made between the main chain and side chain functional groups, and 75 are made between side chains. If we include the water molecules, the hydrogen bonds interactions rise up to 191 interactions. <ref>Betzel, Christian, Gour P. PAL, and Wolfram SAENGER. "Three‐dimensional structure of proteinase K at 0.15‐nm resolution." The FEBS Journal 178.1 (1988): 155-171. Available: https://onlinelibrary.wiley.com/doi/abs/10.1111/j.1432-1033.1988.tb14440.x</ref>. | ||
| - | |||
| - | ---- | ||
| - | ---- | ||
'''Calcium binding sites''' | '''Calcium binding sites''' | ||
| Line 74: | Line 73: | ||
| - | ---- | ||
| - | ---- | ||
| - | + | ||
| + | ===Other structural highlights=== | ||
*The N-terminal and C-terminal are close to each other by interactions made by the Thr16 and Asp260, and by Arg12 and Asp260, Asp187 <ref>Betzel, Christian, Gour P. PAL, and Wolfram SAENGER. "Three‐dimensional structure of proteinase K at 0.15‐nm resolution." The FEBS Journal 178.1 (1988): 155-171. Available: https://onlinelibrary.wiley.com/doi/abs/10.1111/j.1432-1033.1988.tb14440.x</ref>, which can be viewed <scene name='78/786632/C_and_n_terminal/1'>here</scene>. | *The N-terminal and C-terminal are close to each other by interactions made by the Thr16 and Asp260, and by Arg12 and Asp260, Asp187 <ref>Betzel, Christian, Gour P. PAL, and Wolfram SAENGER. "Three‐dimensional structure of proteinase K at 0.15‐nm resolution." The FEBS Journal 178.1 (1988): 155-171. Available: https://onlinelibrary.wiley.com/doi/abs/10.1111/j.1432-1033.1988.tb14440.x</ref>, which can be viewed <scene name='78/786632/C_and_n_terminal/1'>here</scene>. | ||
Revision as of 00:32, 18 June 2018
==Proteinase K==
| |||||||||||
References
- ↑ PDB 2ID8: Wang, Jiawei, Miroslawa Dauter, and Zbigniew Dauter. "What can be done with a good crystal and an accurate beamline?." Acta Crystallographica Section D: Biological Crystallography 62.12 (2006): 1475-1483. Available: https://www.ncbi.nlm.nih.gov/pubmed/17139083
- ↑ Betzel, Christian, Gour P. PAL, and Wolfram SAENGER. "Three‐dimensional structure of proteinase K at 0.15‐nm resolution." The FEBS Journal 178.1 (1988): 155-171. Available: https://onlinelibrary.wiley.com/doi/abs/10.1111/j.1432-1033.1988.tb14440.x
- ↑ Betzel, Christian, Gour P. PAL, and Wolfram SAENGER. "Three‐dimensional structure of proteinase K at 0.15‐nm resolution." The FEBS Journal 178.1 (1988): 155-171. Available: https://onlinelibrary.wiley.com/doi/abs/10.1111/j.1432-1033.1988.tb14440.x
- ↑ Betzel, Christian, Gour P. PAL, and Wolfram SAENGER. "Three‐dimensional structure of proteinase K at 0.15‐nm resolution." The FEBS Journal 178.1 (1988): 155-171. Available: https://onlinelibrary.wiley.com/doi/abs/10.1111/j.1432-1033.1988.tb14440.x
- ↑ Betzel, Christian, Gour P. PAL, and Wolfram SAENGER. "Three‐dimensional structure of proteinase K at 0.15‐nm resolution." The FEBS Journal 178.1 (1988): 155-171. Available: https://onlinelibrary.wiley.com/doi/abs/10.1111/j.1432-1033.1988.tb14440.x
- ↑ Betzel, Christian, Gour P. PAL, and Wolfram SAENGER. "Three‐dimensional structure of proteinase K at 0.15‐nm resolution." The FEBS Journal 178.1 (1988): 155-171. Available: https://onlinelibrary.wiley.com/doi/abs/10.1111/j.1432-1033.1988.tb14440.x
- ↑ Betzel, Christian, Gour P. PAL, and Wolfram SAENGER. "Three‐dimensional structure of proteinase K at 0.15‐nm resolution." The FEBS Journal 178.1 (1988): 155-171. Available: https://onlinelibrary.wiley.com/doi/abs/10.1111/j.1432-1033.1988.tb14440.x
- ↑ Betzel, Christian, Gour P. PAL, and Wolfram SAENGER. "Three‐dimensional structure of proteinase K at 0.15‐nm resolution." The FEBS Journal 178.1 (1988): 155-171. Available: https://onlinelibrary.wiley.com/doi/abs/10.1111/j.1432-1033.1988.tb14440.x
- ↑ Betzel, Christian, Gour P. PAL, and Wolfram SAENGER. "Three‐dimensional structure of proteinase K at 0.15‐nm resolution." The FEBS Journal 178.1 (1988): 155-171. Available: https://onlinelibrary.wiley.com/doi/abs/10.1111/j.1432-1033.1988.tb14440.x
- ↑ Betzel, Christian, Gour P. PAL, and Wolfram SAENGER. "Three‐dimensional structure of proteinase K at 0.15‐nm resolution." The FEBS Journal 178.1 (1988): 155-171. Available: https://onlinelibrary.wiley.com/doi/abs/10.1111/j.1432-1033.1988.tb14440.x
- ↑ Betzel, Christian, Gour P. PAL, and Wolfram SAENGER. "Three‐dimensional structure of proteinase K at 0.15‐nm resolution." The FEBS Journal 178.1 (1988): 155-171. Available: https://onlinelibrary.wiley.com/doi/abs/10.1111/j.1432-1033.1988.tb14440.x
- ↑ Betzel, Christian, Gour P. PAL, and Wolfram SAENGER. "Three‐dimensional structure of proteinase K at 0.15‐nm resolution." The FEBS Journal 178.1 (1988): 155-171. Available: https://onlinelibrary.wiley.com/doi/abs/10.1111/j.1432-1033.1988.tb14440.x
- ↑ Betzel, Christian, Gour P. PAL, and Wolfram SAENGER. "Three‐dimensional structure of proteinase K at 0.15‐nm resolution." The FEBS Journal 178.1 (1988): 155-171. Available: https://onlinelibrary.wiley.com/doi/abs/10.1111/j.1432-1033.1988.tb14440.x
- ↑ Betzel, Christian, Gour P. PAL, and Wolfram SAENGER. "Three‐dimensional structure of proteinase K at 0.15‐nm resolution." The FEBS Journal 178.1 (1988): 155-171. Available: https://onlinelibrary.wiley.com/doi/abs/10.1111/j.1432-1033.1988.tb14440.x
- ↑ Cronier, Sabrina, et al. "Detection and characterization of proteinase K-sensitive disease-related prion protein with thermolysin." Biochemical Journal 416.2 (2008): 297-305. Available online: http://www.biochemj.org/content/416/2/297