This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
User:Ashley Crotteau/Sandbox1
From Proteopedia
(Difference between revisions)
| Line 18: | Line 18: | ||
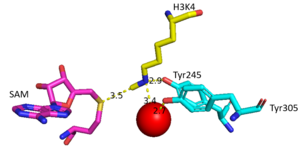
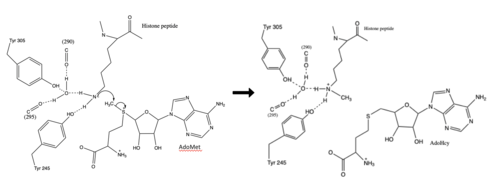
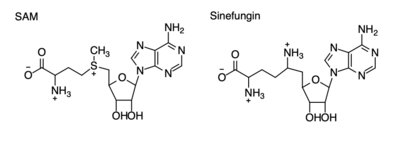
The SET domain is mostly defined by <scene name='81/811707/Variable_knot/2'>turns and loops</scene> with the few <scene name='81/811707/Beta_sheets/3'>antiparallel β-sheets</scene>.<ref name="Schubert" /> <scene name='81/811707/Beta-hairpin/3'>Residues 337-349</scene> form a β-hairpin that sticks out at a right angle to the surface of the enzyme.<ref name="Xiao" /> The following three residues (<scene name='81/811707/Sharp_bend/3'>350-352</scene>) accommodate a sharp bend in the peptide chain and the end of the protein takes on an <scene name='81/811707/C-term_alpha_helix/2'>α-helical conformation</scene>.<ref name="Xiao" /> The two most defining features of the SET domain are the C-terminal tyrosine and the knot-like fold. These two components have been recognized to be essential for S-adenosyl-L-methionine(SAM) binding and catalysis, which is shown as <scene name='81/811707/Sam_isolated/2'>S-adenosyl-L-homocysteine</scene>(SAH) in the structure after methylation of the histone.<ref name="Schubert" /> <ref name="Yeates" /> <ref name="Huang" /> The knot-like fold contains the binding sites for the cofactor SAM and the peptide substrate.<ref name="Licciardello" /> | The SET domain is mostly defined by <scene name='81/811707/Variable_knot/2'>turns and loops</scene> with the few <scene name='81/811707/Beta_sheets/3'>antiparallel β-sheets</scene>.<ref name="Schubert" /> <scene name='81/811707/Beta-hairpin/3'>Residues 337-349</scene> form a β-hairpin that sticks out at a right angle to the surface of the enzyme.<ref name="Xiao" /> The following three residues (<scene name='81/811707/Sharp_bend/3'>350-352</scene>) accommodate a sharp bend in the peptide chain and the end of the protein takes on an <scene name='81/811707/C-term_alpha_helix/2'>α-helical conformation</scene>.<ref name="Xiao" /> The two most defining features of the SET domain are the C-terminal tyrosine and the knot-like fold. These two components have been recognized to be essential for S-adenosyl-L-methionine(SAM) binding and catalysis, which is shown as <scene name='81/811707/Sam_isolated/2'>S-adenosyl-L-homocysteine</scene>(SAH) in the structure after methylation of the histone.<ref name="Schubert" /> <ref name="Yeates" /> <ref name="Huang" /> The knot-like fold contains the binding sites for the cofactor SAM and the peptide substrate.<ref name="Licciardello" /> | ||
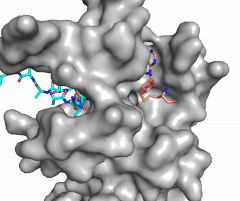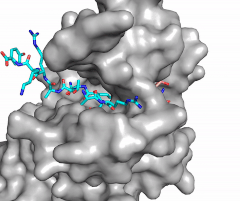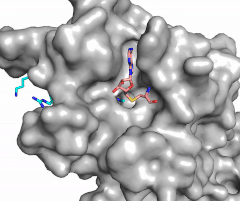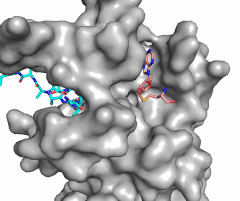
| - | [[Image:Methyl1.gif|300 px| | + | [[Image:Methyl1.gif|300 px|right|thumb|Figure 1: Image of substrate bound on one side of SET7/9 (PDB 1o9s) with the lysine target in the active site channel (cyan) and S-adenosyl homocysteine (pink) bound on the opposite face of the enzyme]] |
===Active Site and Channel=== | ===Active Site and Channel=== | ||
Revision as of 19:05, 24 April 2019
H. sapiens Lysine Methyltransferase, SET 7/9
| |||||||||||
References
[3] [8] [5] [6] [7] [9] [10] [11] [1] [2] [4]
- ↑ 1.0 1.1 DesJarlais R, Tummino PJ. Role of Histone-Modifying Enzymes and Their Complexes in Regulation of Chromatin Biology. Biochemistry. 2016 Mar 22;55(11):1584-99. doi: 10.1021/acs.biochem.5b01210. Epub , 2016 Jan 26. PMID:26745824 doi:http://dx.doi.org/10.1021/acs.biochem.5b01210
- ↑ 2.0 2.1 Marino-Ramirez L, Kann MG, Shoemaker BA, Landsman D. Histone structure and nucleosome stability. Expert Rev Proteomics. 2005 Oct;2(5):719-29. PMID:16209651 doi:http://dx.doi.org/10.1586/14789450.2.5.719
- ↑ 3.00 3.01 3.02 3.03 3.04 3.05 3.06 3.07 3.08 3.09 3.10 Xiao B, Jing C, Wilson JR, Walker PA, Vasisht N, Kelly G, Howell S, Taylor IA, Blackburn GM, Gamblin SJ. Structure and catalytic mechanism of the human histone methyltransferase SET7/9. Nature. 2003 Feb 6;421(6923):652-6. Epub 2003 Jan 22. PMID:12540855 doi:10.1038/nature01378
- ↑ 4.0 4.1 4.2 doi: https://dx.doi.org/10.15406/mojcsr.2016.03.00047
- ↑ 5.0 5.1 5.2 5.3 5.4 5.5 5.6 Schubert HL, Blumenthal RM, Cheng X. Many paths to methyltransfer: a chronicle of convergence. Trends Biochem Sci. 2003 Jun;28(6):329-35. PMID:12826405
- ↑ 6.0 6.1 6.2 6.3 6.4 6.5 Yeates TO. Structures of SET domain proteins: protein lysine methyltransferases make their mark. Cell. 2002 Oct 4;111(1):5-7. PMID:12372294
- ↑ 7.0 7.1 Huang S, Shao G, Liu L. The PR domain of the Rb-binding zinc finger protein RIZ1 is a protein binding interface and is related to the SET domain functioning in chromatin-mediated gene expression. J Biol Chem. 1998 Jun 26;273(26):15933-9. PMID:9632640
- ↑ 8.0 8.1 doi: https://dx.doi.org/10.1016/C2014-0-02189-2
- ↑ 9.0 9.1 9.2 9.3 9.4 9.5 Del Rizzo PA, Couture JF, Dirk LM, Strunk BS, Roiko MS, Brunzelle JS, Houtz RL, Trievel RC. SET7/9 catalytic mutants reveal the role of active site water molecules in lysine multiple methylation. J Biol Chem. 2010 Oct 8;285(41):31849-58. Epub 2010 Aug 1. PMID:20675860 doi:http://dx.doi.org/10.1074/jbc.M110.114587
- ↑ 10.0 10.1 10.2 10.3 10.4 Sun G, Reddy MA, Yuan H, Lanting L, Kato M, Natarajan R. Epigenetic histone methylation modulates fibrotic gene expression. J Am Soc Nephrol. 2010 Dec;21(12):2069-80. doi: 10.1681/ASN.2010060633. Epub 2010, Oct 7. PMID:20930066 doi:http://dx.doi.org/10.1681/ASN.2010060633
- ↑ 11.0 11.1 Tian X, Zhang S, Liu HM, Zhang YB, Blair CA, Mercola D, Sassone-Corsi P, Zi X. Histone lysine-specific methyltransferases and demethylases in carcinogenesis: new targets for cancer therapy and prevention. Curr Cancer Drug Targets. 2013 Jun;13(5):558-79. doi:, 10.2174/1568009611313050007. PMID:23713993 doi:http://dx.doi.org/10.2174/1568009611313050007
Student Contributors
Ashley Crotteau
Parker Hiday
Lauren Allman