This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Sandbox reserved 1560
From Proteopedia
| Line 1: | Line 1: | ||
| + | {{Sandbox_Reserved_BHall_Chem351_F19}}<!-- PLEASE ADD YOUR CONTENT BELOW HERE --> | ||
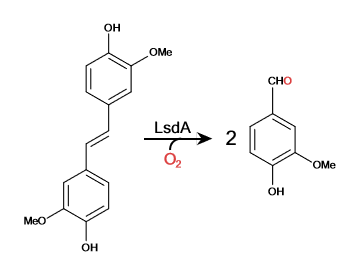
==Lignostilbene-α,β-dioxygenase A (LsdA) Catalyzation== | ==Lignostilbene-α,β-dioxygenase A (LsdA) Catalyzation== | ||
<StructureSection load='6ojt' size='340' side='right' caption='Crystal Structure of Sphingomonas paucimobilis apo-LsdA' scene=''> | <StructureSection load='6ojt' size='340' side='right' caption='Crystal Structure of Sphingomonas paucimobilis apo-LsdA' scene=''> | ||
Revision as of 16:35, 2 December 2019
| This Sandbox is Reserved from Aug 26 through Dec 12, 2019 for use in the course CHEM 351 Biochemistry taught by Bonnie_Hall at the Grand View University, Des Moines, USA. This reservation includes Sandbox Reserved 1556 through Sandbox Reserved 1575. |
To get started:
More help: Help:Editing |
Lignostilbene-α,β-dioxygenase A (LsdA) Catalyzation
| |||||||||||
References
- ↑ Kuatsjah E, Verstraete MM, Kobylarz MJ, Liu AKN, Murphy MEP, Eltis LD. Identification of functionally important residues and structural features in a bacterial lignostilbene dioxygenase. J Biol Chem. 2019 Jul 10. pii: RA119.009428. doi: 10.1074/jbc.RA119.009428. PMID:31292192 doi:http://dx.doi.org/10.1074/jbc.RA119.009428
- ↑ https://pubchem.ncbi.nlm.nih.gov/compound/Vanillin
- ↑ Carroll, Andrew; Somerville, Chris (June 2009). "Cellulosic Biofuels". Annual Review of Plant Biology. 60 (1): 165–182. doi:10.1146/annurev.arplant.043008.092125.<ref></ref> Lignostilbene is the second most abundant raw material.
Energy Transformation
When Phe 59 is substituted with His and Tyr 101 is substituted with Phe, the k (app/cat) decreases in activity by 15-10 folds. The Lys 134 substitution of Met inhibits the enzyme activity. Phenylazophenol inhibits the LsdA-catalyzed cleavage of lignostilbene with a competitive and uncompetitive inhibition.
Structural highlights
and .