This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
User:Abbey Wells/Sandbox 1
From Proteopedia
(Difference between revisions)
| Line 3: | Line 3: | ||
==Introduction== | ==Introduction== | ||
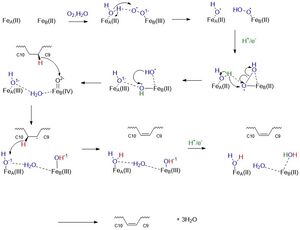
| - | Stearoyl-CoA Desaturase is an enzyme essential for the biosynthesis of monosaturated fatty acids from saturated fatty acids.SCD catalyzes the rate-limiting step in the conversion of [http://en.wikipedia.org/wiki/Stearoyl-CoA Stearoyl-CoA] to [http://en.wikipedia.org/wiki/Oleic_acid oleic acid], an essential substrate in the biosynthesis of phospholipids, triacyclglycerols, and cholesterol. SCD is highly conserved in eukaryotes and has different gene isoforms. Mice have four isoforms: SCD1, SCD2, SCD3, and SCD4. Humans have two different isoforms: SCD1 and SCD5. The SCD discussed in this page is [http://en.wikipedia.org/wiki/Stearoyl-CoA_desaturase-1 Stearoyl-CoA Desaturase 1 (SCD1)] found in mice. SCD is believed to have once been an anaerobic pathway found in cartilaginous fish about 450 million years ago<ref name="Filipe">doi: 10.1186/1471-2148-11-132</ref>. The enzyme’s mechanism is now aerobic and this aerobic pathway is favored. The structure of SCD1 was found using X-ray crystallography. | + | Stearoyl-CoA Desaturase is an enzyme essential for the biosynthesis of monosaturated fatty acids from saturated fatty acids.SCD catalyzes the rate-limiting step in the conversion of [http://en.wikipedia.org/wiki/Stearoyl-CoA Stearoyl-CoA] to [http://en.wikipedia.org/wiki/Oleic_acid oleic acid], an essential substrate in the biosynthesis of phospholipids, triacyclglycerols, and cholesterol <ref name="Bai">PMID:26098370</ref>. SCD is highly conserved in eukaryotes and has different gene isoforms. Mice have four isoforms: SCD1, SCD2, SCD3, and SCD4. Humans have two different isoforms: SCD1 and SCD5. The SCD discussed in this page is [http://en.wikipedia.org/wiki/Stearoyl-CoA_desaturase-1 Stearoyl-CoA Desaturase 1 (SCD1)] found in mice. SCD is believed to have once been an anaerobic pathway found in cartilaginous fish about 450 million years ago<ref name="Filipe">doi: 10.1186/1471-2148-11-132</ref>. The enzyme’s mechanism is now aerobic and this aerobic pathway is favored. The structure of SCD1 was found using X-ray crystallography. |
== Structure == | == Structure == | ||
=== Overall Structure === | === Overall Structure === | ||
| - | SCD1 is an integral membrane protein embedded within the [http://micro.magnet.fsu.edu/cells/endoplasmicreticulum/endoplasmicreticulum.html endoplasmic reticulum] and consists of 4 transmembrane alpha helices, arranged in a cone-like shape. The cytosolic domain of the enzyme consists of 11 alpha helices and contains the carboxy and amino termini. Its substrate, Stearoyl-CoA, binds to the cytosolic region which contains a "kink" that properly orients Stearoyl-CoA to undergo a [http://en.wikipedia.org/wiki/Dehydrogenation#:~:text=Dehydrogenation%20is%20the%20a%20chemical,reaction%20and%20a%20serious%20problem.&text=Enzymes%20that%20catalyze%20dehydrogenation%20are%20called%20dehydrogenases. dehydrogenation] reaction between the <scene name='87/877602/C9_and_c10/1'>Carbon 9 and Carbon 10</scene> of Stearoyl-CoA. | + | SCD1 is an integral membrane protein embedded within the [http://micro.magnet.fsu.edu/cells/endoplasmicreticulum/endoplasmicreticulum.html endoplasmic reticulum] and consists of 4 transmembrane alpha helices, arranged in a cone-like shape. The cytosolic domain of the enzyme consists of 11 alpha helices and contains the carboxy and amino termini <ref name="Bai">PMID:26098370</ref>. Its substrate, Stearoyl-CoA, binds to the cytosolic region which contains a "kink" that properly orients Stearoyl-CoA to undergo a [http://en.wikipedia.org/wiki/Dehydrogenation#:~:text=Dehydrogenation%20is%20the%20a%20chemical,reaction%20and%20a%20serious%20problem.&text=Enzymes%20that%20catalyze%20dehydrogenation%20are%20called%20dehydrogenases. dehydrogenation] reaction between the <scene name='87/877602/C9_and_c10/1'>Carbon 9 and Carbon 10</scene> of Stearoyl-CoA. |
=== Binding of Substrate === | === Binding of Substrate === | ||
Revision as of 16:53, 17 April 2021
Stearoyl-CoA Desaturase 1 from Mus musculus
| |||||||||||
References
- ↑ 1.0 1.1 1.2 1.3 Bai Y, McCoy JG, Levin EJ, Sobrado P, Rajashankar KR, Fox BG, Zhou M. X-ray structure of a mammalian stearoyl-CoA desaturase. Nature. 2015 Jun 22. doi: 10.1038/nature14549. PMID:26098370 doi:http://dx.doi.org/10.1038/nature14549
- ↑ 2.0 2.1 Castro LF, Wilson JM, Goncalves O, Galante-Oliveira S, Rocha E, Cunha I. The evolutionary history of the stearoyl-CoA desaturase gene family in vertebrates. BMC Evol Biol. 2011 May 19;11:132. doi: 10.1186/1471-2148-11-132. PMID:21595943 doi:http://dx.doi.org/10.1186/1471-2148-11-132
- ↑ Shen J, Wu G, Tsai AL, Zhou M. Structure and Mechanism of a Unique Diiron Center in Mammalian Stearoyl-CoA Desaturase. J Mol Biol. 2020 May 27. pii: S0022-2836(20)30367-3. doi:, 10.1016/j.jmb.2020.05.017. PMID:32470559 doi:http://dx.doi.org/10.1016/j.jmb.2020.05.017
Student Contributors
- Abbey Wells
- Josey McKinley
- Anthony Durand