This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Sandbox Reserved 1652
From Proteopedia
(Difference between revisions)
| Line 1: | Line 1: | ||
| + | |||
{{Sandbox_Reserved_ESBS20_}}<!-- PLEASE ADD YOUR CONTENT BELOW HERE --> | {{Sandbox_Reserved_ESBS20_}}<!-- PLEASE ADD YOUR CONTENT BELOW HERE --> | ||
== The Transient Receptor Potential cation channel subfamily V member 1 TRPV1 == | == The Transient Receptor Potential cation channel subfamily V member 1 TRPV1 == | ||
| Line 6: | Line 7: | ||
== Introduction == | == Introduction == | ||
| - | [https://en.wikipedia.org/wiki/TRPV1 TRPV1] (Vanilloid Transient Receptor Potential Type 1) is a non-selective ion channel which, in response to a stimulus, induces an incoming current of cations, primarily calcium and sodium, that causes depolarization of the cell. It is part of the [https://en.wikipedia.org/wiki/Transient_receptor_potential_channel TRP] (Transient Receptor Potential) superfamily and is the first in a subfamily of vanilloid-sensitive TRP channels: TRPVs. | + | [https://en.wikipedia.org/wiki/TRPV1 TRPV1] (Vanilloid Transient Receptor Potential Type 1) is a non-selective ion channel which, in response to a stimulus, induces an incoming current of cations, primarily calcium and sodium, that causes depolarization of the cell. It is part of the [https://en.wikipedia.org/wiki/Transient_receptor_potential_channel TRP] (Transient Receptor Potential) superfamily and is the first in a subfamily of vanilloid-sensitive TRP channels: TRPVs. |
| - | + | This receptor is expressed by sensory neurons of the dorsal and trigeminal spinal ganglia.TRPV1 is implicated in [https://en.wikipedia.org/wiki/Nociception nociception], its activation by heat or by chemical substances leads to a painful sensation.<ref name="TRPV1">Wikipedia contributors. (2020b, décembre 21). TRPV1. Wikipedia. https://en.wikipedia.org/wiki/TRPV1 (Consulté le: déc. 28, 2020). [En ligne].</ref> | |
| - | + | ||
== Structure of TRPV1 == | == Structure of TRPV1 == | ||
| Line 14: | Line 14: | ||
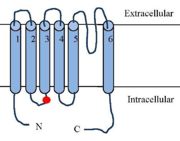
[[Image:TRPV1 Struct.jpg| thumb| Schematic figure of the TRPV1 receptor]] | [[Image:TRPV1 Struct.jpg| thumb| Schematic figure of the TRPV1 receptor]] | ||
| - | The TRPV1 receptor is a transmembrane protein receptor. It is made up of '''839 amino acids'''. It’s molecular weight is '''94 938Da''' | + | The TRPV1 receptor is a transmembrane protein receptor. It is made up of '''839 amino acids'''. It’s molecular weight is '''94 938Da'''.<ref name="Structure of the TRPV1 ion channel determined by electron cryo-microscopy">Liao, M., Cao, E., Julius, D., & Cheng, Y. (2013b). Structure of the TRPV1 ion channel determined by electron cryo-microscopy. Nature, 504(7478), 107‑112. https://doi.org/10.1038/nature12822(consulté le déc. 28, 2020)</ref> TRPV1 exists in two states : the open state and the closed state.<ref> T. Rosenbaum et S. A. Simon, « TRPV1 Receptors and Signal Transduction », in TRP Ion Channel Function in Sensory Transduction and Cellular Signaling Cascades, W. B. Liedtke et S. Heller, Éd. Boca Raton (FL): CRC Press/Taylor & Francis, 2007</ref> |
TRPV1 are '''tetrameric''' channel type receptors. The four subunits form a symmetry plane around a pore allowing the passage of ions. | TRPV1 are '''tetrameric''' channel type receptors. The four subunits form a symmetry plane around a pore allowing the passage of ions. | ||
Each TRPV1 subunits is made of one '''N-terminal tail''', one '''transmembrane region''', a '''C-terminal tail''' preceded by a '''TRP domain'''. The N-terminal and C-terminal region are intracellular. N and C terminal region are responsible of 70% of the total mass of TRPV1.<ref name="Structure of the TRPV1 ion channel determined by electron cryo-microscopy"/> | Each TRPV1 subunits is made of one '''N-terminal tail''', one '''transmembrane region''', a '''C-terminal tail''' preceded by a '''TRP domain'''. The N-terminal and C-terminal region are intracellular. N and C terminal region are responsible of 70% of the total mass of TRPV1.<ref name="Structure of the TRPV1 ion channel determined by electron cryo-microscopy"/> | ||
| - | The<scene name='86/868185/Aterm/1'> N-terminal</scene> | + | The<scene name='86/868185/Aterm/1'> N-terminal</scene> region has 6 repeats of [https://en.wikipedia.org/wiki/Ankyrin <scene name='86/868185/Ankyrin_residues_of_n-term/1'>ankyrin</scene>].<ref name="Structure of the TRPV1 ion channel determined by electron cryo-microscopy"/><ref name="Integrating TRPV1 Receptor Function with Capsaicin Psychophysics">G. Smutzer et R. K. Devassy, « Integrating TRPV1 Receptor Function with Capsaicin Psychophysics », Advances in Pharmacological Sciences, janv. 14, 2016</ref> |
The transmembrane region is composed of '''six transmembrane a helices''' (<scene name='86/868185/S1/1'>S1</scene>,<scene name='86/868185/S2/1'>S2</scene>,<scene name='86/868185/S3/1'>S3</scene>,<scene name='86/868185/S4/1'>S4</scene>,<scene name='86/868185/S5/1'>S5</scene>,<scene name='86/868185/S6/1'>S6</scene>). S1,S2 and S3 helices contain aromatic side chain (<scene name='86/868185/Y511_s512_t550/2'>Y511,S512,T550</scene>).<ref name="Structure of the TRPV1 ion channel determined by electron cryo-microscopy"/> A small hydrophobic domain beetween S5 and S6 with a <scene name='86/868185/Re_entrant_loop/1'>re-entrant loop</scene> constitutes the pore allowing the passage of ions through the TRPV1 receptor.<ref name="TRPV1"/><ref name="Structure of the TRPV1 ion channel determined by electron cryo-microscopy"/> | The transmembrane region is composed of '''six transmembrane a helices''' (<scene name='86/868185/S1/1'>S1</scene>,<scene name='86/868185/S2/1'>S2</scene>,<scene name='86/868185/S3/1'>S3</scene>,<scene name='86/868185/S4/1'>S4</scene>,<scene name='86/868185/S5/1'>S5</scene>,<scene name='86/868185/S6/1'>S6</scene>). S1,S2 and S3 helices contain aromatic side chain (<scene name='86/868185/Y511_s512_t550/2'>Y511,S512,T550</scene>).<ref name="Structure of the TRPV1 ion channel determined by electron cryo-microscopy"/> A small hydrophobic domain beetween S5 and S6 with a <scene name='86/868185/Re_entrant_loop/1'>re-entrant loop</scene> constitutes the pore allowing the passage of ions through the TRPV1 receptor.<ref name="TRPV1"/><ref name="Structure of the TRPV1 ion channel determined by electron cryo-microscopy"/> | ||
| Line 24: | Line 24: | ||
The S6 domain links the receptor to the <scene name='86/868185/C_term/1'>C-terminal</scene> domain of TRPV1. The C-terminal is made of 150 amino acids and it contains '''<scene name='86/868185/Trp/1'>TRP domain</scene>'''.<ref name="Integrating TRPV1 Receptor Function with Capsaicin Psychophysics"/>.The TRP domain is made of 23-25 aminoacids with a alpha helical structure, it is found in many TRP family members.<ref name="Structure of the TRPV1 ion channel determined by electron cryo-microscopy"/> TRP domain is necessary for the formation of tetrameric TRPV1. | The S6 domain links the receptor to the <scene name='86/868185/C_term/1'>C-terminal</scene> domain of TRPV1. The C-terminal is made of 150 amino acids and it contains '''<scene name='86/868185/Trp/1'>TRP domain</scene>'''.<ref name="Integrating TRPV1 Receptor Function with Capsaicin Psychophysics"/>.The TRP domain is made of 23-25 aminoacids with a alpha helical structure, it is found in many TRP family members.<ref name="Structure of the TRPV1 ion channel determined by electron cryo-microscopy"/> TRP domain is necessary for the formation of tetrameric TRPV1. | ||
| - | Many amino-acids of the C-terminal domain are the target of post-translationnal modifications by [https://en.wikipedia.org/wiki/Kinase kinases] and [https://en.wikipedia.org/wiki/Phosphatase phosphatases].<ref>X. Yao, H.-Y. Kwan, et Y. Huang, « Regulation of TRP Channels by Phosphorylation | + | Many amino-acids of the C-terminal domain are the target of post-translationnal modifications by [https://en.wikipedia.org/wiki/Kinase kinases] and [https://en.wikipedia.org/wiki/Phosphatase phosphatases].<ref>X. Yao, H.-Y. Kwan, et Y. Huang, « Regulation of TRP Channels by Phosphorylation », Neurosignals, vol. 14, no 6, p. 273‑280, 2005, doi: 10.1159/000093042</ref> |
== Relation structure-function == | == Relation structure-function == | ||
| Line 36: | Line 36: | ||
[[Image:128px-Capsaicin Formulae.png | thumb|Chemical structure of capsaicin]] | [[Image:128px-Capsaicin Formulae.png | thumb|Chemical structure of capsaicin]] | ||
[https://en.wikipedia.org/wiki/Capsaicin Capsaicin] is an active compound in chili. | [https://en.wikipedia.org/wiki/Capsaicin Capsaicin] is an active compound in chili. | ||
| - | TRPV1 receptor has a '''capsaicin-binding pocket'' formed by S3,S4 and <scene name='86/868185/S4s5_linker/1'>S4-S5 linker</scene>. The capsaicin-binding pocket is surrounded by the residues <scene name='86/868185/Y511_s512_t550/2'>Y511,S512,T550</scene>.<ref>F. Yang et J. Zheng, « Understand spiciness: mechanism of TRPV1 channel activation by capsaicin », Protein Cell, vol. 8, no 3, p. 169‑177, mars 2017, doi: 10.1007/s13238-016-0353-7.</ref> | + | TRPV1 receptor has a '''capsaicin-binding pocket''' formed by S3,S4 and <scene name='86/868185/S4s5_linker/1'>S4-S5 linker</scene>. The capsaicin-binding pocket is surrounded by the residues <scene name='86/868185/Y511_s512_t550/2'>Y511,S512,T550</scene>.<ref>F. Yang et J. Zheng, « Understand spiciness: mechanism of TRPV1 channel activation by capsaicin », Protein Cell, vol. 8, no 3, p. 169‑177, mars 2017, doi: 10.1007/s13238-016-0353-7.</ref> |
Bound capsaicin is oriented in a « tail-up, head down » configuration. In this configuration,capsaicin is anchored into the receptor.<ref>F. Yang et al., « Structural mechanism underlying capsaicin binding and activation of the TRPV1 ion channel », Nat. Chem. Biol., vol. 11, no 7, Art. no 7, juill. 2015, doi: 10.1038/nchembio.1835.</ref> | Bound capsaicin is oriented in a « tail-up, head down » configuration. In this configuration,capsaicin is anchored into the receptor.<ref>F. Yang et al., « Structural mechanism underlying capsaicin binding and activation of the TRPV1 ion channel », Nat. Chem. Biol., vol. 11, no 7, Art. no 7, juill. 2015, doi: 10.1038/nchembio.1835.</ref> | ||
| - | The capsaicin cycle binds via hydrogen bounds to amino acids on the S3 helix (<scene name='86/868185/Y511/1'>Y511</scene>), on the <scene name='86/868185/S4s5_linker/2'>S4-S5 linker</scene> and on the S6 helix (<scene name='86/868185/Tyr671/1'>T671</scene>). The amid group of capsaicin binds the <scene name='86/868185/S4/2'>S4</scene> helix.<ref name="Integrating TRPV1 Receptor Function with Capsaicin Psychophysics | + | The capsaicin cycle binds via hydrogen bounds to amino acids on the S3 helix (<scene name='86/868185/Y511/1'>Y511</scene>), on the <scene name='86/868185/S4s5_linker/2'>S4-S5 linker</scene> and on the S6 helix (<scene name='86/868185/Tyr671/1'>T671</scene>). The amid group of capsaicin binds the <scene name='86/868185/S4/2'>S4</scene> helix.<ref name="Integrating TRPV1 Receptor Function with Capsaicin Psychophysics"> |
Capsaicin maintains TRPV1 in an open state by «pull and contact» interactions. A conformational change wave spread over the whole pore.<ref>F. Yang et al., « The conformational wave in capsaicin activation of transient receptor potential vanilloid 1 ion channel », Nat. Commun., vol. 9, no 1, Art. no 1, juill. 2018, doi: 10.1038/s41467-018-05339-6.</ref>. | Capsaicin maintains TRPV1 in an open state by «pull and contact» interactions. A conformational change wave spread over the whole pore.<ref>F. Yang et al., « The conformational wave in capsaicin activation of transient receptor potential vanilloid 1 ion channel », Nat. Commun., vol. 9, no 1, Art. no 1, juill. 2018, doi: 10.1038/s41467-018-05339-6.</ref>. | ||
| - | Capsaicin maintains TRPV1 in an open state. A conformational change wave spread over the whole pore.<ref>F. Yang et al., « The conformational wave in capsaicin activation of transient receptor potential vanilloid 1 ion channel », Nat. Commun., vol. 9, no 1, Art. no 1, juill. 2018, doi: 10.1038/s41467-018-05339-6.</ref>. This leads to the massive enter of Ca2+ and Na+ and the depolarization of the nerve fiber. Depolarization triggers the generation of an [https://en.wikipedia.org/wiki/Action_potential action potential] causing a painful sensation.<ref name="TRPV1"/ | + | Capsaicin maintains TRPV1 in an open state. A conformational change wave spread over the whole pore.<ref>F. Yang et al., « The conformational wave in capsaicin activation of transient receptor potential vanilloid 1 ion channel », Nat. Commun., vol. 9, no 1, Art. no 1, juill. 2018, doi: 10.1038/s41467-018-05339-6.</ref>. This leads to the massive enter of Ca2+ and Na+ and the depolarization of the nerve fiber. Depolarization triggers the generation of an [https://en.wikipedia.org/wiki/Action_potential action potential] causing a painful sensation.<ref name="TRPV1"/> |
| - | + | ||
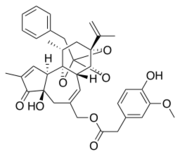
====Resiniferatoxin (RTX)==== | ====Resiniferatoxin (RTX)==== | ||
| Line 60: | Line 59: | ||
====Sensitization==== | ====Sensitization==== | ||
| - | '''Phosphorylation''' of the TRPV1 receptor leads to its sensitization | + | '''Phosphorylation''' of the TRPV1 receptor leads to its sensitization. Phosphorylations are either caused by '''PKC''' ([https://en.wikipedia.org/wiki/Inositol_trisphosphate IP3 signalling]), by '''PKA''' ([https://fr.wikipedia.org/wiki/Adénylate_cyclase AMPc signalling]), or by '''CamKII'''.<ref>K. W. Ho, N. J. Ward, et D. J. Calkins, « TRPV1: a stress response protein in the central nervous system », Am. J. Neurodegener. Dis., vol. 1, no 1, p. 1‑14, avr. 2012.</ref><ref name="Integrating TRPV1 Receptor Function with Capsaicin Psychophysics">.PKA phosphorylates <scene name='86/868185/S502_t370/1'>T370 and S502</scene>, PKC and CaMKII phosphorylate <scene name='86/868185/Ser502_thr704/1'>S502 and T704</scene>. |
| - | + | ||
The phosphorylation of TRPV1 lead to an increase in the expression of TRPV1 at the membrane surface.<ref>K. W. Ho, N. J. Ward, et D. J. Calkins, « TRPV1: a stress response protein in the central nervous system », Am. J. Neurodegener. Dis., vol. 1, no 1, p. 1‑14, avr. 2012.</ref>. The phosphorylation of TRPV1 lead to an '''over-expression ''' of TRPV1 at the membrane surface.<ref>K. W. Ho, N. J. Ward, et D. J. Calkins, « TRPV1: a stress response protein in the central nervous system », Am. J. Neurodegener. Dis., vol. 1, no 1, p. 1‑14, avr. 2012.</ref> Moreover, phosphorylated TRPV1 would have a reduced channel opening threshold.<ref>G. Bhave et al., « Protein kinase C phosphorylation sensitizes but does not activate the capsaicin receptor transient receptor potential vanilloid 1 (TRPV1) », Proc. Natl. Acad. Sci., vol. 100, no 21, p. 12480‑12485, oct. 2003, doi: 10.1073/pnas.2032100100.</ref>. As a result phosphorylated TRPV1 are more responsive to agonist and the resulting pain sensation is higher. | The phosphorylation of TRPV1 lead to an increase in the expression of TRPV1 at the membrane surface.<ref>K. W. Ho, N. J. Ward, et D. J. Calkins, « TRPV1: a stress response protein in the central nervous system », Am. J. Neurodegener. Dis., vol. 1, no 1, p. 1‑14, avr. 2012.</ref>. The phosphorylation of TRPV1 lead to an '''over-expression ''' of TRPV1 at the membrane surface.<ref>K. W. Ho, N. J. Ward, et D. J. Calkins, « TRPV1: a stress response protein in the central nervous system », Am. J. Neurodegener. Dis., vol. 1, no 1, p. 1‑14, avr. 2012.</ref> Moreover, phosphorylated TRPV1 would have a reduced channel opening threshold.<ref>G. Bhave et al., « Protein kinase C phosphorylation sensitizes but does not activate the capsaicin receptor transient receptor potential vanilloid 1 (TRPV1) », Proc. Natl. Acad. Sci., vol. 100, no 21, p. 12480‑12485, oct. 2003, doi: 10.1073/pnas.2032100100.</ref>. As a result phosphorylated TRPV1 are more responsive to agonist and the resulting pain sensation is higher. | ||
| Line 68: | Line 66: | ||
====Desensitization==== | ====Desensitization==== | ||
| - | A repeated exposure of TRPV1 to capsaicin fails to activate the receptor. It occurs by a Ca2+-dependent mechanism that leads to a ''' | + | A repeated exposure of TRPV1 to capsaicin fails to activate the receptor. It occurs by a Ca2+-dependent mechanism that leads to a '''desphosphorylation''' by the '''calcineurin''' [https://en.wikipedia.org/wiki/Phosphatase phosphatase] of the serine and threonine residues which have been previously phosphorylated by PKA (<scene name='86/868185/S502_t370/2'>S502 and T370</scene>). Thus, the decrease in TRPV1 phosphorylation diminish the sensitivity of the capsaicin channel and leads to a decrease in capsaicin's response by '''negative feedback'''. |
The '''over-stimulation''' of TRPV1 is followed by the nerve endings' death due to calcium overload, causing analgesia. <ref name="Integrating TRPV1 Receptor Function with Capsaicin Psychophysics">G. Smutzer et R. K. Devassy, « Integrating TRPV1 Receptor Function with Capsaicin Psychophysics », Advances in Pharmacological Sciences, janv. 14, 2016</ref> | The '''over-stimulation''' of TRPV1 is followed by the nerve endings' death due to calcium overload, causing analgesia. <ref name="Integrating TRPV1 Receptor Function with Capsaicin Psychophysics">G. Smutzer et R. K. Devassy, « Integrating TRPV1 Receptor Function with Capsaicin Psychophysics », Advances in Pharmacological Sciences, janv. 14, 2016</ref> | ||
| Line 74: | Line 72: | ||
== Implication of TRPV1 in the treatment of pain == | == Implication of TRPV1 in the treatment of pain == | ||
| - | + | In 2011 Qutenza (NeurogesX) patch containing 8% of capsaicin has been markered in France and indicated in the [https://en.wikipedia.org/wiki/Neuropathic_pain neuropathic pain]. | |
| - | + | ||
| - | + | ||
| - | + | ||
| - | In 2011 Qutenza (NeurogesX) patch containing 8% of capsaicin has been | + | |
The absorption through the skin of these creams generated partial desensitization of the nerve endings. This is the cause of a decrease in painful sensations.<ref name="TRPV1 dans les neuropathies douloureuses - Des modèles animaux aux perspectives thérapeutiques">A. Danigo, L. Magy, et C. Demiot, « TRPV1 dans les neuropathies douloureuses - Des modèles animaux aux perspectives thérapeutiques », médecine/sciences, vol. 29, no 6‑7, Art. no 6‑7, juin 2013, doi: 10.1051/medsci/2013296012.</ref> | The absorption through the skin of these creams generated partial desensitization of the nerve endings. This is the cause of a decrease in painful sensations.<ref name="TRPV1 dans les neuropathies douloureuses - Des modèles animaux aux perspectives thérapeutiques">A. Danigo, L. Magy, et C. Demiot, « TRPV1 dans les neuropathies douloureuses - Des modèles animaux aux perspectives thérapeutiques », médecine/sciences, vol. 29, no 6‑7, Art. no 6‑7, juin 2013, doi: 10.1051/medsci/2013296012.</ref> | ||
| - | + | ||
| - | Many laboratories are conducting clinical studies on oral TRPV1 antagonists: | + | Many laboratories are conducting clinical studies on oral TRPV1 antagonists: GlaxoSmithKline, Amgen, Merk-Neurogen, Abbot, Eli-Lilly-Glenmark, AstraZeneca and Japan Tobacco. The major problem with these pain relievers is the [https://en.wikipedia.org/wiki/Hyperthermia hyperthermia] generated in humans. These effects caused these studies to be stopped in phase I.<ref name="TRPV1 dans les neuropathies douloureuses - Des modèles animaux aux perspectives thérapeutiques"/> |
| - | + | ||
| - | + | ||
| - | + | ||
Revision as of 09:27, 30 December 2021
| This Sandbox is Reserved from 26/11/2020, through 26/11/2021 for use in the course "Structural Biology" taught by Bruno Kieffer at the University of Strasbourg, ESBS. This reservation includes Sandbox Reserved 1643 through Sandbox Reserved 1664. |
To get started:
More help: Help:Editing |
The Transient Receptor Potential cation channel subfamily V member 1 TRPV1
| |||||||||||
References
- ↑ 1.0 1.1 1.2 Wikipedia contributors. (2020b, décembre 21). TRPV1. Wikipedia. https://en.wikipedia.org/wiki/TRPV1 (Consulté le: déc. 28, 2020). [En ligne].
- ↑ 2.0 2.1 2.2 2.3 2.4 2.5 Liao, M., Cao, E., Julius, D., & Cheng, Y. (2013b). Structure of the TRPV1 ion channel determined by electron cryo-microscopy. Nature, 504(7478), 107‑112. https://doi.org/10.1038/nature12822(consulté le déc. 28, 2020)
- ↑ T. Rosenbaum et S. A. Simon, « TRPV1 Receptors and Signal Transduction », in TRP Ion Channel Function in Sensory Transduction and Cellular Signaling Cascades, W. B. Liedtke et S. Heller, Éd. Boca Raton (FL): CRC Press/Taylor & Francis, 2007
- ↑ 4.0 4.1 4.2 4.3 4.4 G. Smutzer et R. K. Devassy, « Integrating TRPV1 Receptor Function with Capsaicin Psychophysics », Advances in Pharmacological Sciences, janv. 14, 2016
- ↑ R. Kumar, A. Hazan, A. Basu, N. Zalcman, H. Matzner, et A. Priel, « Tyrosine Residue in the TRPV1 Vanilloid Binding Pocket Regulates Deactivation Kinetics », J. Biol. Chem., vol. 291, no 26, p. 13855‑13863, juin 2016, doi: 10.1074/jbc.M116.726372.
- ↑ X. Yao, H.-Y. Kwan, et Y. Huang, « Regulation of TRP Channels by Phosphorylation », Neurosignals, vol. 14, no 6, p. 273‑280, 2005, doi: 10.1159/000093042
- ↑ F. Yang et J. Zheng, « Understand spiciness: mechanism of TRPV1 channel activation by capsaicin », Protein Cell, vol. 8, no 3, p. 169‑177, mars 2017, doi: 10.1007/s13238-016-0353-7.
- ↑ F. Yang et al., « Structural mechanism underlying capsaicin binding and activation of the TRPV1 ion channel », Nat. Chem. Biol., vol. 11, no 7, Art. no 7, juill. 2015, doi: 10.1038/nchembio.1835.
- ↑ F. Yang et al., « The conformational wave in capsaicin activation of transient receptor potential vanilloid 1 ion channel », Nat. Commun., vol. 9, no 1, Art. no 1, juill. 2018, doi: 10.1038/s41467-018-05339-6.
- ↑ 10.0 10.1 K. Elokely et al., « Understanding TRPV1 activation by ligands: Insights from the binding modes of capsaicin and resiniferatoxin », Proc. Natl. Acad. Sci., vol. 113, no 2, p. E137‑E145, janv. 2016, doi:10.1073/pnas.1517288113.
- ↑ K. W. Ho, N. J. Ward, et D. J. Calkins, « TRPV1: a stress response protein in the central nervous system », Am. J. Neurodegener. Dis., vol. 1, no 1, p. 1‑14, avr. 2012.
- ↑ K. W. Ho, N. J. Ward, et D. J. Calkins, « TRPV1: a stress response protein in the central nervous system », Am. J. Neurodegener. Dis., vol. 1, no 1, p. 1‑14, avr. 2012.
- ↑ G. Bhave et al., « Protein kinase C phosphorylation sensitizes but does not activate the capsaicin receptor transient receptor potential vanilloid 1 (TRPV1) », Proc. Natl. Acad. Sci., vol. 100, no 21, p. 12480‑12485, oct. 2003, doi: 10.1073/pnas.2032100100.
- ↑ 14.0 14.1 A. Danigo, L. Magy, et C. Demiot, « TRPV1 dans les neuropathies douloureuses - Des modèles animaux aux perspectives thérapeutiques », médecine/sciences, vol. 29, no 6‑7, Art. no 6‑7, juin 2013, doi: 10.1051/medsci/2013296012.