This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Sandbox Reserved 1710
From Proteopedia
(Difference between revisions)
| Line 20: | Line 20: | ||
== Function == | == Function == | ||
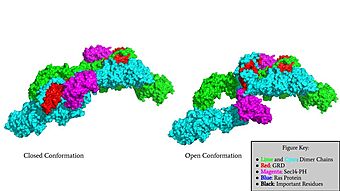
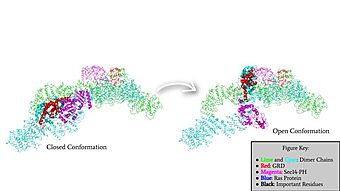
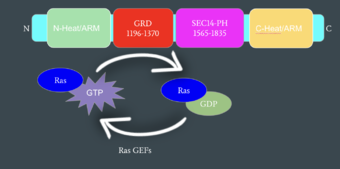
| - | Neurofibromin functions as a tumor suppressor protein.<ref name="Trovó-Marqui">PMID:16813595</ref> Its job is to prevent cell growth by turning off another protein known as [https://en.wikipedia.org/wiki/Ras_GTPase Ras] which in its active state, stimulates cell growth and division. Ras is a GTPase membrane protein that can only interact with Neurofibromin, a cytoplasmic protein, in the open conformation of Neurofibromin. As Neurofibromin is a cytoplasmic protein, it is brought to the membrane to associate with Ras via another protein known as [https://medlineplus.gov/genetics/gene/spred1/ SPRED1]. Neurofibromin can interact with SPRED1 in both the open and closed conformations however, it can only associate with Ras when it is in its open conformation. The interaction between Neurofibromin and Ras occurs via an | + | Neurofibromin functions as a tumor suppressor protein.<ref name="Trovó-Marqui">PMID:16813595</ref> Its job is to prevent cell growth by turning off another protein known as [https://en.wikipedia.org/wiki/Ras_GTPase Ras] which in its active state, stimulates cell growth and division. Ras is a GTPase membrane protein that can only interact with Neurofibromin, a cytoplasmic protein, in the open conformation of Neurofibromin. As Neurofibromin is a cytoplasmic protein, it is brought to the membrane to associate with Ras via another protein known as [https://medlineplus.gov/genetics/gene/spred1/ SPRED1]. Neurofibromin can interact with SPRED1 in both the open and closed conformations however, it can only associate with Ras when it is in its open conformation. The interaction between Neurofibromin and Ras occurs via an Arginine finger (R1276) present in the GRD domain of Neurofibromin which is critical for Ras binding. R1276 is only accessible for binding when the GRD and Sec14-PH domains are rotated into the open conformation and there is no steric hindrance from the surrounding dimer chains. When R1276 is able to associate with Ras, Neurofibromin downregulates the [https://en.wikipedia.org/wiki/MAPK/ERK_pathway Ras signaling pathway] by hydrolyzing the GTP associated with Ras to GDP, effectively making it inactive and inhibiting cell growth and division. |
[[Image:mechanismofRas.png|340 px|right|thumb|Mechanism of Ras Regulation by Neurofibromin]] | [[Image:mechanismofRas.png|340 px|right|thumb|Mechanism of Ras Regulation by Neurofibromin]] | ||
== Disease and Medical Relevance == | == Disease and Medical Relevance == | ||
| - | Currently, there are over 1485 mutations of Neurofibromin that have been identified. Mutations in Neurofibromin can lead to life-threatening illnesses or conditions such as Neurofibromatosis Type I due to the inability of Neurofibromin to interact with Ras.<ref name="Lupton">PMID: 34887559 </ref> The role of Neurofibromin is to inhibit cellular proliferation via its | + | Currently, there are over 1485 mutations of Neurofibromin that have been identified. Mutations in Neurofibromin can lead to life-threatening illnesses or conditions such as Neurofibromatosis Type I due to the inability of Neurofibromin to interact with Ras.<ref name="Lupton">PMID: 34887559 </ref> The role of Neurofibromin is to inhibit cellular proliferation via its Guanine triphosphatase-activating protein (GAP) activity; therefore, when there are mutations to the NF1 gene that prevent the interaction of Neufibromin with Ras, there is nothing stopping Ras from promoting cell growth. Uncontrolled cell growth can lead to tumors and a higher risk of cancer.<ref name="Ratner">PMID:25877329</ref> [https://medlineplus.gov/genetics/condition/neurofibromatosis-type-1/ Neurofibromatosis Type 1], the most well-known disease resulting from mutations in Neurofibromin, is characterized by cognitive impairment, soft, non-cancerous tumors on or under the skin known as neurofibromas, birthmarks called cafe-au-lait macules, clusters of freckles in unusual places, and problems with the bones, eyes and nervous system. |
| - | Neurofibromin is an essential protein and is involved mainly in the differentiation of neural crest derived cells, mesenchymal cells, neural cells, melanocytes, and bone cells. As Neurofibromin is essential for embryonic development, mutations to the NF1 gene can result in psychological retardation resulting from Type I neurofibromatosis. Most of the 1485 mutations identified lead to a synthesis of truncated, non-functional protein and are a result of point mutations. Type I Neurofibromatosis is inherited in autosomal dominant manner but about 50% of cases de novo ones. <ref name="Abramowicz">PMID:25182393</ref> | + | Neurofibromin is an essential protein and is involved mainly in the differentiation of neural crest-derived cells, mesenchymal cells, neural cells, melanocytes, and bone cells. As Neurofibromin is essential for embryonic development, mutations to the NF1 gene can result in psychological retardation resulting from Type I neurofibromatosis. Most of the 1485 mutations identified lead to a synthesis of truncated, non-functional protein and are a result of point mutations. Type I Neurofibromatosis is inherited in an autosomal dominant manner but about 50% of cases de novo ones. <ref name="Abramowicz">PMID:25182393</ref> |
== Student Contributors == | == Student Contributors == | ||
Revision as of 01:12, 29 March 2022
| This Sandbox is Reserved from February 28 through September 1, 2022 for use in the course CH462 Biochemistry II taught by R. Jeremy Johnson at the Butler University, Indianapolis, USA. This reservation includes Sandbox Reserved 1700 through Sandbox Reserved 1729. |
To get started:
More help: Help:Editing |
Human Neurofibromin - The Tumor Suppressor Gene
| |||||||||||
References
- ↑ Naschberger A, Baradaran R, Rupp B, Carroni M. The structure of neurofibromin isoform 2 reveals different functional states. Nature. 2021 Nov;599(7884):315-319. doi: 10.1038/s41586-021-04024-x. Epub 2021, Oct 27. PMID:34707296 doi:http://dx.doi.org/10.1038/s41586-021-04024-x
- ↑ Trovo-Marqui AB, Tajara EH. Neurofibromin: a general outlook. Clin Genet. 2006 Jul;70(1):1-13. doi: 10.1111/j.1399-0004.2006.00639.x. PMID:16813595 doi:http://dx.doi.org/10.1111/j.1399-0004.2006.00639.x
- ↑ Lupton CJ, Bayly-Jones C, D'Andrea L, Huang C, Schittenhelm RB, Venugopal H, Whisstock JC, Halls ML, Ellisdon AM. The cryo-EM structure of the human neurofibromin dimer reveals the molecular basis for neurofibromatosis type 1. Nat Struct Mol Biol. 2021 Dec;28(12):982-988. doi: 10.1038/s41594-021-00687-2., Epub 2021 Dec 9. PMID:34887559 doi:http://dx.doi.org/10.1038/s41594-021-00687-2
- ↑ Ratner N, Miller SJ. A RASopathy gene commonly mutated in cancer: the neurofibromatosis type 1 tumour suppressor. Nat Rev Cancer. 2015 May;15(5):290-301. doi: 10.1038/nrc3911. Epub 2015 Apr 16. PMID:25877329 doi:http://dx.doi.org/10.1038/nrc3911
- ↑ Abramowicz A, Gos M. Neurofibromin in neurofibromatosis type 1 - mutations in NF1gene as a cause of disease. Dev Period Med. 2014 Jul-Sep;18(3):297-306. PMID:25182393