User:George G. Papadeas/Sandbox VKOR
From Proteopedia
(Difference between revisions)
| Line 6: | Line 6: | ||
== Introduction== | == Introduction== | ||
=== Biological Role === | === Biological Role === | ||
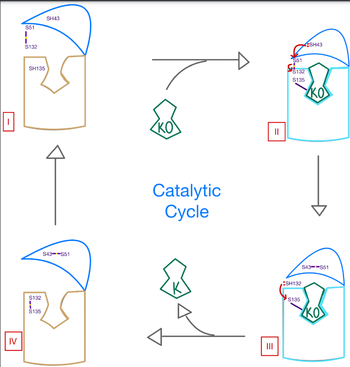
| - | <scene name='90/906893/Vkor_structure/1'>Vitamin K epoxide reductase</scene> (VKOR) is a reducing enzyme composed of 4-helices that spans the endoplasmic reticulum as a transmembrane protein <ref>DOI 10.1126</ref>. Its enzymatic role is reducing <scene name='90/906893/Vkor_with_ko/1'>vitamin K epoxide</scene> (KO) to [https://en.wikipedia.org/wiki/Vitamin_K Vitamin K] Vitamin K hydroquinone (KH2) (Figure 1). | + | <scene name='90/906893/Vkor_structure/1'>Vitamin K epoxide reductase</scene> (VKOR) is a reducing enzyme composed of 4-helices that spans the endoplasmic reticulum as a transmembrane protein <ref>DOI 10.1126</ref>. Its enzymatic role is reducing <scene name='90/906893/Vkor_with_ko/1'>vitamin K epoxide</scene> (KO) to [https://en.wikipedia.org/wiki/Vitamin_K Vitamin K] Vitamin K hydroquinone (KH2) (Figure 1). The mechanism first occurs through the binding KO and using two cysteine residues to reduce KO into Vitamin K. Then, a second pair of cysteine residues will reduce Vitamin K into the final product, KH2 (Figure 1). One of VKORs primary roles is to assist in blood coagulation through this KH2 regeneration mechanism. With Vitamin K as a cofactor, the [https://www.britannica.com/science/bleeding/The-extrinsic-pathway-of-blood-coagulation#ref64617 γ-carboxylase] enzyme will enact post-translational modification on KH2, oxidizing it back to KO. The oxidation of KH2 by γ-carboxylase is coupled with the carboxylation of a glutamate residue to form γ-carboxyglutamate. The coupling of this oxidation and carboxylation will activate several clotting factors in the coagulation cascade. |
| - | + | ||
=== Author's Notes === | === Author's Notes === | ||
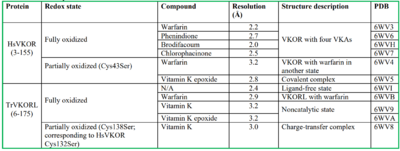
| - | Crystal structures of VKOR were captured with a bound substrate (KO) or vitamin K antagonist (VKA) (PDB Codes: Table 1)<ref>DOI 10.1126</ref>. VKA substrates utilized were anticoagulants, namely [https://en.wikipedia.org/wiki/Warfarin Warfarin], [https://en.wikipedia.org/wiki/Brodifacoum Brodifacoum], [https://en.wikipedia.org/wiki/Phenindione Phenindione], and [https://en.wikipedia.org/wiki/Chlorophacinone Chlorophacinone]. Second, VKOR-like homologs were utilized to aid in structure classification. Homologs refer to specific cysteine residues that have been mutated to serine to facilitate capturing a stable conformation state. Homologs were mainly isolated from human VKOR with some isolated from the pufferfish ''Takifugu rubripes''. Furthermore, all of the structures used have been processed to remove a beta barrel at the south end of VKOR that served no purpose in function of the enzyme. This also allowed for the residue numbering to be reassigned and more closely replicate the human VKOR. | + | Structural characterization of VKOR has been difficult due to its in vitro instability. Recently, a series of atomic structures have been determined utilizing anticoagulant stabilization and VKOR-like [https://pubmed.ncbi.nlm.nih.gov/33154105/ homologs]. Crystal structures of VKOR were captured with a bound substrate (KO) or vitamin K antagonist (VKA) (PDB Codes: Table 1)<ref>DOI 10.1126</ref>. VKA substrates utilized were anticoagulants, namely [https://en.wikipedia.org/wiki/Warfarin Warfarin], [https://en.wikipedia.org/wiki/Brodifacoum Brodifacoum], [https://en.wikipedia.org/wiki/Phenindione Phenindione], and [https://en.wikipedia.org/wiki/Chlorophacinone Chlorophacinone]. Second, VKOR-like homologs were utilized to aid in structure classification. Homologs refer to specific cysteine residues that have been mutated to serine to facilitate capturing a stable conformation state. Homologs were mainly isolated from human VKOR with some isolated from the pufferfish ''Takifugu rubripes''. Furthermore, all of the structures used have been processed to remove a beta barrel at the south end of VKOR that served no purpose in function of the enzyme. This also allowed for the residue numbering to be reassigned and more closely replicate the human VKOR. |
== Structural Highlights== | == Structural Highlights== | ||
Revision as of 15:24, 12 April 2022
VKOR
| |||||||||||
References
1. Li, Weikai et al. “Structure of a bacterial homologue of vitamin K epoxide reductase.” Nature vol. 463,7280 (2010): 507-12. doi:10.1038/nature08720.
2. Liu S, Li S, Shen G, Sukumar N, Krezel AM, Li W. Structural basis of antagonizing the vitamin K catalytic cycle for anticoagulation. Science. 2021 Jan 1;371(6524):eabc5667. doi: 10.1126/science.abc5667. Epub 2020 Nov 5. PMID: 33154105; PMCID: PMC7946407.
- ↑ Hanson, R. M., Prilusky, J., Renjian, Z., Nakane, T. and Sussman, J. L. (2013), JSmol and the Next-Generation Web-Based Representation of 3D Molecular Structure as Applied to Proteopedia. Isr. J. Chem., 53:207-216. doi:http://dx.doi.org/10.1002/ijch.201300024
- ↑ Herraez A. Biomolecules in the computer: Jmol to the rescue. Biochem Mol Biol Educ. 2006 Jul;34(4):255-61. doi: 10.1002/bmb.2006.494034042644. PMID:21638687 doi:10.1002/bmb.2006.494034042644
- ↑ Unknown PubmedID 10.1126
- ↑ Unknown PubmedID 10.1126