This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Sandbox Reserved 1767
From Proteopedia
(Difference between revisions)
| Line 23: | Line 23: | ||
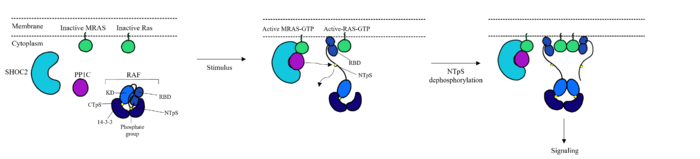
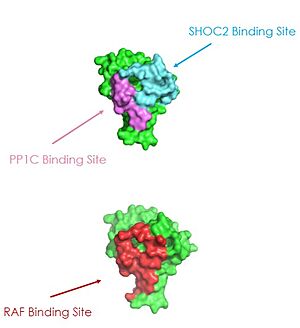
PP1C binds to SHOC2 through a hydrophobic N-terminal disordered region that is complimentary to the <scene name='95/952695/Rvxf_motif/2'>RVXF Motif on SHOC2</scene> and adjacent to a catalytic metal ions <ref name="Liau">PMID: 35768504</ref>. In the RAS/RAF signaling cascade, the region of RAF that is C-terminal to the phosphate group binds to this hydrophobic groove, and the remaining residues bind to the hydrophobic region of SHOC2 <ref name="Hauseman">PMID:35830882</ref>. RAF binding to this region of SHOC2 is what allows PP1C to be specific when in the SMP complex in comparison to PP1C on its own <ref name="Hauseman">PMID:35830882</ref>. Similarly to SHOC2, PP1C does not undergo a <scene name='95/952694/Pp1coverlay/4'>significant conformational change</scene> when SHOC2 and MRAS-GTP bind. The lack of conformational change shows that the structure of PP1C is not dependent on the SMP complex, but in order to act as a phosphatase it must be bound to the complex <ref name="Liau">PMID: 35768504</ref>. | PP1C binds to SHOC2 through a hydrophobic N-terminal disordered region that is complimentary to the <scene name='95/952695/Rvxf_motif/2'>RVXF Motif on SHOC2</scene> and adjacent to a catalytic metal ions <ref name="Liau">PMID: 35768504</ref>. In the RAS/RAF signaling cascade, the region of RAF that is C-terminal to the phosphate group binds to this hydrophobic groove, and the remaining residues bind to the hydrophobic region of SHOC2 <ref name="Hauseman">PMID:35830882</ref>. RAF binding to this region of SHOC2 is what allows PP1C to be specific when in the SMP complex in comparison to PP1C on its own <ref name="Hauseman">PMID:35830882</ref>. Similarly to SHOC2, PP1C does not undergo a <scene name='95/952694/Pp1coverlay/4'>significant conformational change</scene> when SHOC2 and MRAS-GTP bind. The lack of conformational change shows that the structure of PP1C is not dependent on the SMP complex, but in order to act as a phosphatase it must be bound to the complex <ref name="Liau">PMID: 35768504</ref>. | ||
| - | PP1C is involved in many different cellular signaling pathways including [https://www.ncbi.nlm.nih.gov/books/NBK545161/. protein synthesis], [https://www.ncbi.nlm.nih.gov/books/NBK559006/. muscle contraction,] and even carbohydrate metabolism<ref name="Kelker">PMID: 18992256</ref>. In all these pathways, including the SMP pathway, PP1C does not exist as a monomer, it is present in holoenzyme form complex with one of two regulatory subunits ensuring there is no sporadic pathway activation <ref name="Liau">PMID: 35768504</ref>. | + | PP1C is involved in many different cellular signaling pathways including [https://www.ncbi.nlm.nih.gov/books/NBK545161/. protein synthesis], [https://www.ncbi.nlm.nih.gov/books/NBK559006/. muscle contraction,] and even [https://pubmed.ncbi.nlm.nih.gov/11237211/] carbohydrate metabolism]<ref name="Kelker">PMID: 18992256</ref>. In all these pathways, including the SMP pathway, PP1C does not exist as a monomer, it is present in holoenzyme form complex with one of two regulatory subunits ensuring there is no sporadic pathway activation <ref name="Liau">PMID: 35768504</ref>. |
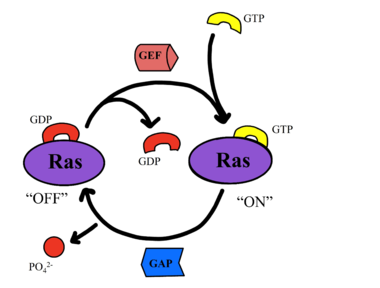
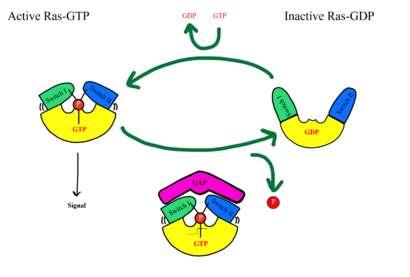
===RAS/RAF === | ===RAS/RAF === | ||
Revision as of 15:38, 17 April 2023
| This Sandbox is Reserved from February 27 through August 31, 2023 for use in the course CH462 Biochemistry II taught by R. Jeremy Johnson at the Butler University, Indianapolis, USA. This reservation includes Sandbox Reserved 1765 through Sandbox Reserved 1795. |
To get started:
More help: Help:Editing |
SHOC2-PP1C-MRAS
| |||||||||||
References
- ↑ 1.0 1.1 1.2 1.3 1.4 1.5 1.6 1.7 Kwon JJ, Hajian B, Bian Y, Young LC, Amor AJ, Fuller JR, Fraley CV, Sykes AM, So J, Pan J, Baker L, Lee SJ, Wheeler DB, Mayhew DL, Persky NS, Yang X, Root DE, Barsotti AM, Stamford AW, Perry CK, Burgin A, McCormick F, Lemke CT, Hahn WC, Aguirre AJ. Structure-function analysis of the SHOC2-MRAS-PP1C holophosphatase complex. Nature. 2022 Jul 13. pii: 10.1038/s41586-022-04928-2. doi:, 10.1038/s41586-022-04928-2. PMID:35831509 doi:http://dx.doi.org/10.1038/s41586-022-04928-2
- ↑ 2.00 2.01 2.02 2.03 2.04 2.05 2.06 2.07 2.08 2.09 2.10 Hauseman ZJ, Fodor M, Dhembi A, Viscomi J, Egli D, Bleu M, Katz S, Park E, Jang DM, Porter KA, Meili F, Guo H, Kerr G, Molle S, Velez-Vega C, Beyer KS, Galli GG, Maira SM, Stams T, Clark K, Eck MJ, Tordella L, Thoma CR, King DA. Structure of the MRAS-SHOC2-PP1C phosphatase complex. Nature. 2022 Jul 13. pii: 10.1038/s41586-022-05086-1. doi:, 10.1038/s41586-022-05086-1. PMID:35830882 doi:http://dx.doi.org/10.1038/s41586-022-05086-1
- ↑ 3.00 3.01 3.02 3.03 3.04 3.05 3.06 3.07 3.08 3.09 3.10 3.11 3.12 3.13 3.14 3.15 3.16 3.17 3.18 3.19 3.20 3.21 3.22 3.23 Liau NPD, Johnson MC, Izadi S, Gerosa L, Hammel M, Bruning JM, Wendorff TJ, Phung W, Hymowitz SG, Sudhamsu J. Structural basis for SHOC2 modulation of RAS signalling. Nature. 2022 Jun 29. pii: 10.1038/s41586-022-04838-3. doi:, 10.1038/s41586-022-04838-3. PMID:35768504 doi:http://dx.doi.org/10.1038/s41586-022-04838-3
- ↑ 4.0 4.1 4.2 4.3 4.4 4.5 4.6 4.7 Lavoie H, Therrien M. Structural keys unlock RAS-MAPK cellular signalling pathway. Nature. 2022 Sep;609(7926):248-249. PMID:35970881 doi:10.1038/d41586-022-02189-7
- ↑ 5.0 5.1 Young LC, Hartig N, Boned Del Río I, Sari S, Ringham-Terry B, Wainwright JR, Jones GG, McCormick F, Rodriguez-Viciana P. SHOC2-MRAS-PP1 complex positively regulates RAF activity and contributes to Noonan syndrome pathogenesis. Proc Natl Acad Sci U S A. 2018 Nov 6;115(45):E10576-E10585. PMID:30348783 doi:10.1073/pnas.1720352115
- ↑ Kelker MS, Page R, Peti W. Crystal structures of protein phosphatase-1 bound to nodularin-R and tautomycin: a novel scaffold for structure-based drug design of serine/threonine phosphatase inhibitors. J Mol Biol. 2009 Jan 9;385(1):11-21. Epub 2008 Nov 1. PMID:18992256 doi:10.1016/j.jmb.2008.10.053
Student Contributors
- Sloan August
- Rosa Trippel
- Kayla Wilhoite