We apologize for Proteopedia being slow to respond. For the past two years, a new implementation of Proteopedia has been being built. Soon, it will replace this 18-year old system. All existing content will be moved to the new system at a date that will be announced here.
1s9v
From Proteopedia
(Difference between revisions)
| Line 15: | Line 15: | ||
<jmolCheckbox> | <jmolCheckbox> | ||
<scriptWhenChecked>; select protein; define ~consurf_to_do selected; consurf_initial_scene = true; script "/wiki/ConSurf/s9/1s9v_consurf.spt"</scriptWhenChecked> | <scriptWhenChecked>; select protein; define ~consurf_to_do selected; consurf_initial_scene = true; script "/wiki/ConSurf/s9/1s9v_consurf.spt"</scriptWhenChecked> | ||
| - | <scriptWhenUnchecked>script /wiki/extensions/Proteopedia/spt/ | + | <scriptWhenUnchecked>script /wiki/extensions/Proteopedia/spt/initialview03.spt</scriptWhenUnchecked> |
<text>to colour the structure by Evolutionary Conservation</text> | <text>to colour the structure by Evolutionary Conservation</text> | ||
</jmolCheckbox> | </jmolCheckbox> | ||
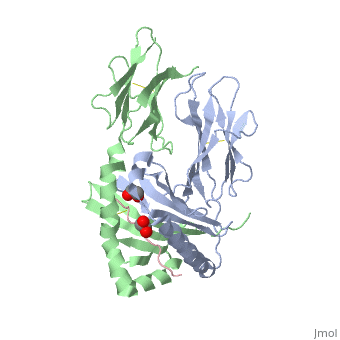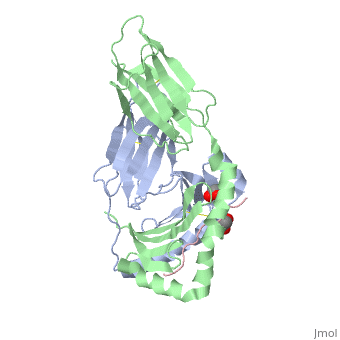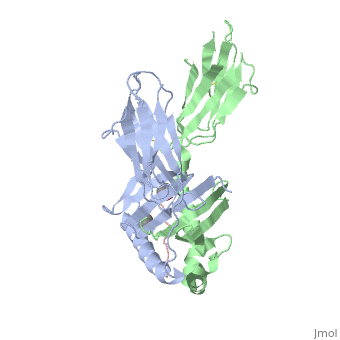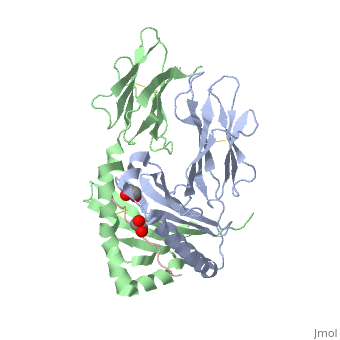
</jmol>, as determined by [http://consurfdb.tau.ac.il/ ConSurfDB]. You may read the [[Conservation%2C_Evolutionary|explanation]] of the method and the full data available from [http://bental.tau.ac.il/new_ConSurfDB/main_output.php?pdb_ID=1s9v ConSurf]. | </jmol>, as determined by [http://consurfdb.tau.ac.il/ ConSurfDB]. You may read the [[Conservation%2C_Evolutionary|explanation]] of the method and the full data available from [http://bental.tau.ac.il/new_ConSurfDB/main_output.php?pdb_ID=1s9v ConSurf]. | ||
<div style="clear:both"></div> | <div style="clear:both"></div> | ||
| + | <div style="background-color:#fffaf0;"> | ||
| + | == Publication Abstract from PubMed == | ||
| + | Celiac disease, also known as celiac sprue, is a gluten-induced autoimmune-like disorder of the small intestine, which is strongly associated with HLA-DQ2. The structure of DQ2 complexed with an immunogenic epitope from gluten, QLQPFPQPELPY, has been determined to 2.2-A resolution by x-ray crystallography. The glutamate at P6, which is formed by tissue transglutaminase-catalyzed deamidation, is an important anchor residue as it participates in an extensive hydrogen-bonding network involving Lys-beta71 of DQ2. The gluten peptide-DQ2 complex retains critical hydrogen bonds between the MHC and the peptide backbone despite the presence of many proline residues in the peptide that are unable to participate in amide-mediated hydrogen bonds. Positioning of proline residues such that they do not interfere with backbone hydrogen bonding results in a reduction in the number of registers available for gluten peptides to bind to MHC class II molecules and presumably impairs the likelihood of establishing favorable side-chain interactions. The HLA association in celiac disease can be explained by a superior ability of DQ2 to bind the biased repertoire of proline-rich gluten peptides that have survived gastrointestinal digestion and that have been deamidated by tissue transglutaminase. Finally, surface-exposed proline residues in the proteolytically resistant ligand were replaced with functionalized analogs, thereby providing a starting point for the design of orally active agents for blocking gluten-induced toxicity. | ||
| + | |||
| + | Structural basis for HLA-DQ2-mediated presentation of gluten epitopes in celiac disease.,Kim CY, Quarsten H, Bergseng E, Khosla C, Sollid LM Proc Natl Acad Sci U S A. 2004 Mar 23;101(12):4175-9. Epub 2004 Mar 12. PMID:15020763<ref>PMID:15020763</ref> | ||
| + | |||
| + | From MEDLINE®/PubMed®, a database of the U.S. National Library of Medicine.<br> | ||
| + | </div> | ||
| + | <div class="pdbe-citations 1s9v" style="background-color:#fffaf0;"></div> | ||
==See Also== | ==See Also== | ||
| Line 25: | Line 34: | ||
*[[MHC 3D structures|MHC 3D structures]] | *[[MHC 3D structures|MHC 3D structures]] | ||
*[[MHC II 3D structures|MHC II 3D structures]] | *[[MHC II 3D structures|MHC II 3D structures]] | ||
| + | == References == | ||
| + | <references/> | ||
__TOC__ | __TOC__ | ||
</StructureSection> | </StructureSection> | ||
Current revision
Crystal structure of HLA-DQ2 complexed with deamidated gliadin peptide
| |||||||||||