User:Erin May/Sandbox 1
From Proteopedia
| Line 16: | Line 16: | ||
===PrP<sup>C</sup> natural monomer=== | ===PrP<sup>C</sup> natural monomer=== | ||
| - | <StructureSection load='1qlx' size='300' side='left' caption='Major Prion Protein [[ | + | <StructureSection load='1qlx' size='300' side='left' caption='Major Prion Protein [[1QLX]]' scene=''> |
Anything in this section will appear adjacent to the 3D structure and will be scrollable. | Anything in this section will appear adjacent to the 3D structure and will be scrollable. | ||
| Line 25: | Line 25: | ||
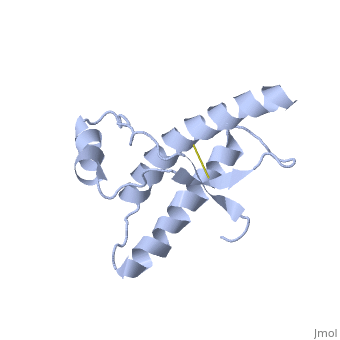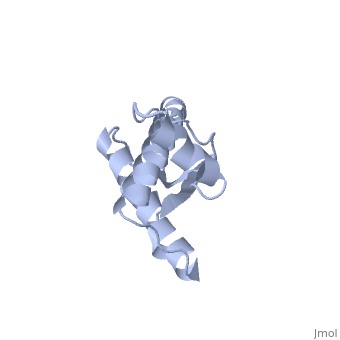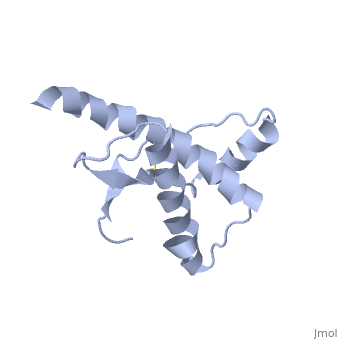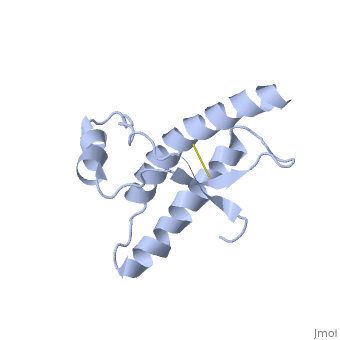
===Dimer Form=== | ===Dimer Form=== | ||
| - | <StructureSection load='1i4m' size='300' side='right' caption='Major Prion Protein: Dimerized [[ | + | <StructureSection load='1i4m' size='300' side='right' caption='Major Prion Protein: Dimerized [[1I4M]]' scene=''> |
Anything in this section will appear adjacent to the 3D structure and will be scrollable. | Anything in this section will appear adjacent to the 3D structure and will be scrollable. | ||
| Line 33: | Line 33: | ||
=== PrP<sup>Sc</sup> | === PrP<sup>Sc</sup> | ||
| + | <StructureSection load='2rnm' size='300' side='right' caption='Amyloid formation: Human Prion Protein [[2RNM]]' scene=''> | ||
| + | |||
| + | Anything in this section will appear adjacent to the 3D structure and will be scrollable. | ||
| + | |||
| + | </StructureSection> | ||
== Reference List == | == Reference List == | ||
{{reflist}} | {{reflist}} | ||
Revision as of 05:02, 27 November 2012
Contents |
Prions as a disease causing agent
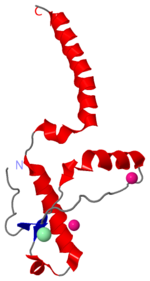
Prions are infectious or genetic misfolded proteins which act as templates upon which properly folded prion protein monomers can aggregate. Prions contain no nucleic acid such as other infectoius molecules or organisms. Human Prion Protein or Major Prion protein, exists as a normal constituent of human cells, found mostly in the brain[2] and is called PrPC.[3] PrPC is composed of mostly helix whereas the infectious form, PrPSc, is composed of high percentage beta sheets.[3]
The diseases prions confer are neurodegenerative disorders which result from the large scale aggregation of these proteins. For more information about the infections related to prions see Transmissible spongiform encephalopathy at Wikipedia.
Unfolding Mechanism
Currently, the mechanism by which a template prion unfolds a the helices of a properly folded prion protein is unknown. Specific residues have been shown to either confer resistance or lend themselves to this unfolding.
PrPC natural monomer
| |||||||||||
Dimer Form
| |||||||||||
=== PrPSc
| |||||||||||