This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
DAHP synthase
From Proteopedia
(Difference between revisions)
| Line 6: | Line 6: | ||
== Structural highlights == | == Structural highlights == | ||
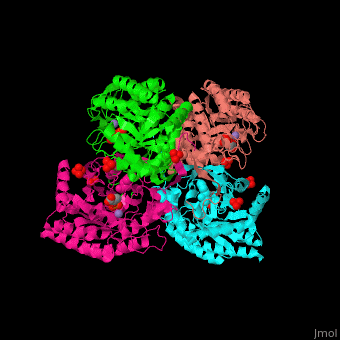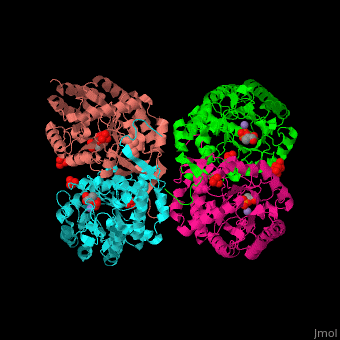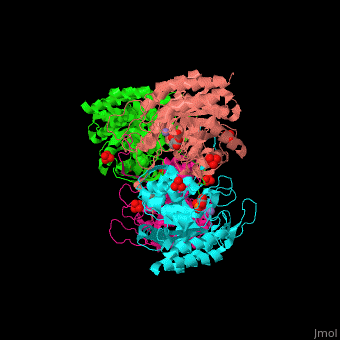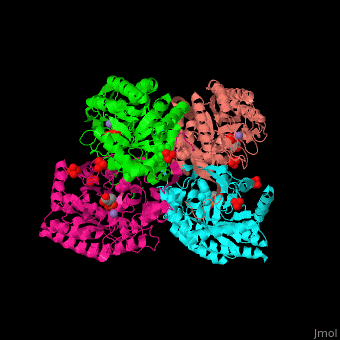
| - | The <scene name='70/708806/Cv/3'>bivalent metal is bound to a Cys-X-X-His motif</scene>. The DAHPS active site is located in a channel at the C-terminal of the enzyme where the substrate (PEP), inhibitor (phenylalanine) and the metal ion (Mn+2) are seen.<ref>PMID:12126632</ref> | + | The <scene name='70/708806/Cv/3'>bivalent metal is bound to a Cys-X-X-His motif</scene>. The DAHPS active site is located in a channel at the C-terminal of the enzyme where the <scene name='70/708806/Cv/4'>substrate (PEP)</scene>, inhibitor (phenylalanine) and the metal ion (Mn+2) are seen.<ref>PMID:12126632</ref> |
</StructureSection> | </StructureSection> | ||
Revision as of 09:02, 28 December 2015
| |||||||||||
3D Structures of DAHP synthase
Updated on 28-December-2015
References
- ↑ Stephens CM, Bauerle R. Analysis of the metal requirement of 3-deoxy-D-arabino-heptulosonate-7-phosphate synthase from Escherichia coli. J Biol Chem. 1991 Nov 5;266(31):20810-7. PMID:1682314
- ↑ Shumilin IA, Zhao C, Bauerle R, Kretsinger RH. Allosteric inhibition of 3-deoxy-D-arabino-heptulosonate-7-phosphate synthase alters the coordination of both substrates. J Mol Biol. 2002 Jul 26;320(5):1147-56. PMID:12126632