This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Sandbox Wabash13
From Proteopedia
(Difference between revisions)
| Line 10: | Line 10: | ||
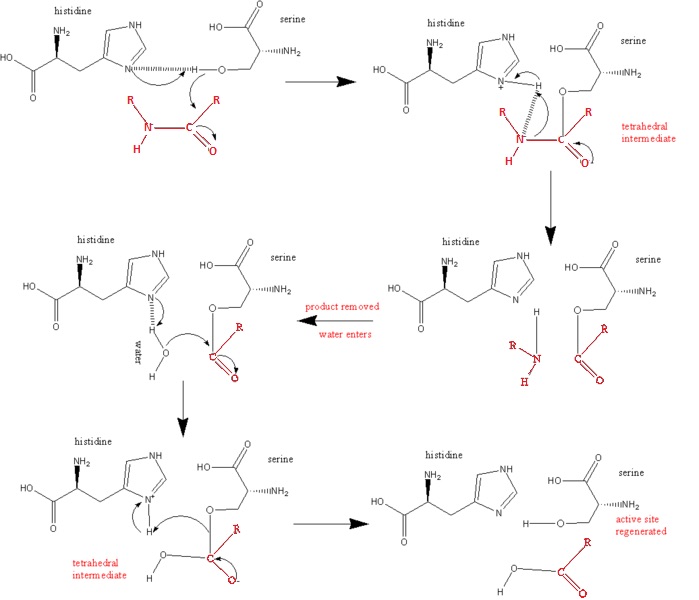
2. Acid catalysis breaks the tetrahedral intermediate through cleaving of the scissile peptide bond to form an acyl-enzyme intermediate. His 57 donates a proton by general acid catalysis. | 2. Acid catalysis breaks the tetrahedral intermediate through cleaving of the scissile peptide bond to form an acyl-enzyme intermediate. His 57 donates a proton by general acid catalysis. | ||
| - | This is aided by | + | This is aided by Asp 102 polarizing effect on His 57. This causes the tetrahedral intermediate to decompose to the acyl-enzyme intermediate. |
| - | 3. The amine product is replaced by H2O and subsequently released from the enzyme/substrate complex. | + | 3. The amine product is replaced by H2O and subsequently released from the enzyme/substrate complex. |
| + | R'NH2 is the new N-nterminal portion of the cleaved polypeptide chain. (See Diagram Below) | ||
| - | 4. Base catalysis by enzyme | + | 4. Base catalysis by enzyme. The Acyl Intermediate,highly susceptible to hydrolytic cleavage, adds water to yield a secondary tetrahedral intermediate. |
| + | H2O forms a covalent bond with the carbonyl group of the N-terminal peptide, leading to another tetrahedral intermediate | ||
| + | |||
| - | 5. Acid catalysis by the breaking of the C-O covalent bond of the tetrahedral intermediate, releasing the peptide from the enzyme substrate complex. Once the peptide is released, the enzyme once again becomes active. <ref>PMID:16636277</ref>. | + | 5. Acid catalysis by the breaking of the C-O covalent bond of the tetrahedral intermediate, releasing the peptide from the enzyme substrate complex. Once the peptide is released, the enzyme once again becomes active. |
| + | By yielding a carboxylate product ( C-Terminal portion of the cleaved polypeptide chain) that regenerates the active enzyme | ||
| + | |||
| + | <ref>PMID:16636277</ref>. | ||
Revision as of 01:37, 19 February 2016
Trypsin Mechanism & Structure - Chase Francoeur, Elias Arellano
| |||||||||||