This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Journal:Molecular Cell:1
From Proteopedia
(Difference between revisions)

| Line 25: | Line 25: | ||
Comparison of the <span style="color:yellow;background-color:black;font-weight:bold;">dAChE4 design model (yellow)</span> with the <span style="color:lime;background-color:black;font-weight:bold;">solved crystal structure (PDB entry: [[5hq3]], green)</span> and <span style="color:violet;background-color:black;font-weight:bold;">(wild-type hAChE (PDB entry: [[4ey4]], violet)</span>: | Comparison of the <span style="color:yellow;background-color:black;font-weight:bold;">dAChE4 design model (yellow)</span> with the <span style="color:lime;background-color:black;font-weight:bold;">solved crystal structure (PDB entry: [[5hq3]], green)</span> and <span style="color:violet;background-color:black;font-weight:bold;">(wild-type hAChE (PDB entry: [[4ey4]], violet)</span>: | ||
| - | *<scene name='72/728277/Cv/14'>Overall | + | *<scene name='72/728277/Cv/14'>Overall alignment</scene>. |
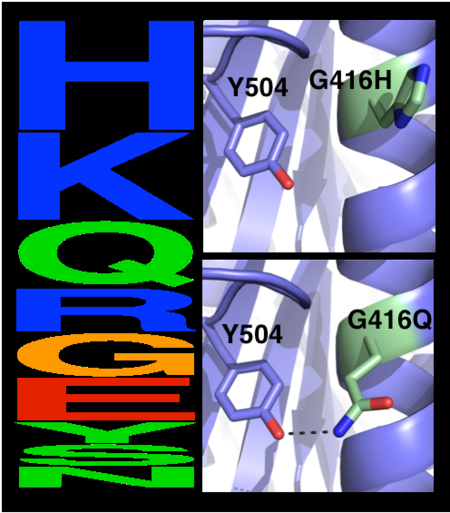
*<scene name='72/728277/Cv/13'>Sub-Ångstrom accuracy in design of 2 small-to-large core mutations</scene>. | *<scene name='72/728277/Cv/13'>Sub-Ångstrom accuracy in design of 2 small-to-large core mutations</scene>. | ||
*<scene name='72/728277/Cv/19'>The maximal deviation observed between any respective Cα atoms</scene> in the model and structure is 3.1 Å (dashed line). This conformation change likely results from elimination of a side chain-backbone hydrogen bond between Thr112 and Ser110 due to the designed Thr112Ala mutation. | *<scene name='72/728277/Cv/19'>The maximal deviation observed between any respective Cα atoms</scene> in the model and structure is 3.1 Å (dashed line). This conformation change likely results from elimination of a side chain-backbone hydrogen bond between Thr112 and Ser110 due to the designed Thr112Ala mutation. | ||
Revision as of 16:20, 3 May 2016
| |||||||||||
- ↑ REF
Proteopedia Page Contributors and Editors (what is this?)
This page complements a publication in scientific journals and is one of the Proteopedia's Interactive 3D Complement pages. For aditional details please see I3DC.