| Colicins are a type of bacteriocin - peptide and protein antibiotics released by bacteria to kill other bacteria of the same species, in order to provide a competitive advantage for nutrient acquisition [1]. Bacteriocins are named after their species of origin; colicins are so-called because they are produced by E. Coli[2]. Because of their narrow killing spectrum which focuses primarily on the species which has made the peptide (or occasionally closely related species[3]), bacteriocins are important in microbial biodiversity and the stable co-existence of the bacterial populations[4][5].
Colicin peptides are plasmid-encoded. The peptide is released by the cell into the area surrounding it, and then parasitises proteins present in the host cell membrane to translocate across into the host cell. Many protein-protein interactions are involved in the cell entry, and the main system is involved in the grouping of colicins into two families: Group A colicins use the Tol system to enter the host cell, and Group B use the Ton system. Once inside the host cell, the cell killing follows 1st order kinetics - ie one molecule is theoretically sufficient to kill the cell[6].
The structure of all colicins, of which over 20 have been identified, follows a 3 domain design:
At the N terminus is the Translocation domain (T-): Residues in ColIa.
The Receptor binding domain is at the centre of the peptide (R-): Residues in ColIa.
The C terminus contains the Cytotoxic domain (C-): Residues in ColIa[7].
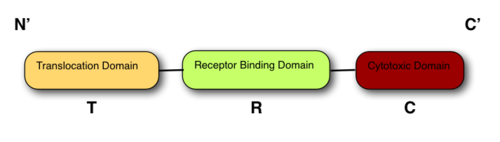 The 3 domain structure of all colicins Some colicins exhibit DNase Activity and others TRNase activity. For more details see also
Synthesis, Production and Release
Synthesis of many colicins is repressed by the LexA protein, which is part of the SOS regulon[8].
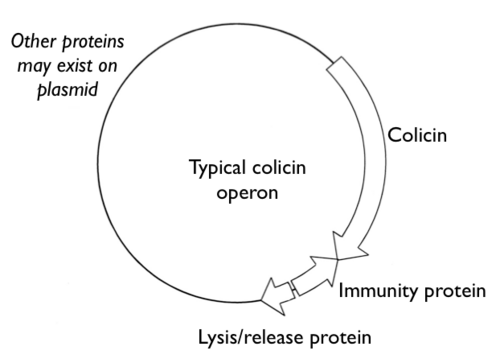 The structure of a typical colicin operon, highlighting the 3 proteins encoded together. Targeting and Receptors
Colicins vary significantly in the receptors that they target to initiate their uptake. The majority of the group A colicins use the BtuB receptor, which is present on E. coli as a vitamin B12 uptake receptor. Once bound to the receptor, the coiled-coil receptor binding domain unfolds, in an essential step that removes the immunity protein and triggers translocation[9]. Other colicins use other receptors - generally involved in the uptake of small metabolite growth factors.
Colicin Uptake
Colicins are divided into two groups depending on the method of uptake which they target. Group A colicins use the Tol system to bind to and enter the target cell, and group B use the Ton system. The Tol system consists of 5 proteins - TolA, TolB, TolR, TolQ and Pal[10], and group A proteins using this often recruit a second co-receptor involved in translocation, usually OmpF or TolC, but could be OmpC and PhoE[11]. The Ton system consists of TonB, ExbB and ExbD[12], and no known co-receptor is utilised in translocation[13].It could be possible that Ton-dependent colicins are indiscriminate in use of coreceptors, or that the colicins move down the outside wall of a β barrel protein[14]. It is known that colicins do unfold during translocation, but the peptides resulting from this exceed the diameter of pores formed by any of the molecules mentioned above[15][16]. However, while unfolding does occur, this is not induced by receptor binding in either Tol or Ton dependent colicins[17].
Understanding how the colicins can cross the membrane is highly important, as if this could be targeted and exploited it could be useful for novel therapeutic agents[18]. It is also estimated that a single colicin molecule is sufficient to kill the bacterial cell, following first order kinetics[19].
Killing Activities
Colicins kill their target cell through a variety of different methods. The main killing activities are carried out through Pore Formation, DNase Activity and 16s rRNase activity, and some colicins also exhibit tRNase activity.
The killing activities carried out by colicins could be used medicinally as an alternative to antibiotics in the case where the specific strain of E. coli can be identified[20], and as potential natural replacements for food preservatives[21].
List of colicins, with their translocation proteins and cytotoxic activity
| Colicin | Group | OM Receptor | Translocation Proteins | Cytotoxic activity | Immunity protein
|
| Colicin A | A | BtuB | OmpF/TolQRAB | Pore-forming | Colicin_Immunity_Protein[22]
|
| Colicin E1 | A | BtuB | TolC/TolAQ | Pore-forming | ImmE1[23]
|
| Colicin E2 | A | BtuB | OmpF/TolQRAB | DNase | Im2[24]
|
| Colicin E3 | A | BtuB | OmpF/TolQRAB | 16s rRNase | Im3[25]
|
| Colicin E4 | A | BtuB | OmpF/TolQRAB | 16s rRNase | Im4[26]
|
| Colicin E5 | A | BtuB | OmpF/TolQRAB | tRNase | ImmE5[27]
|
| Colicin E6 | A | BtuB | OmpF/TolQRAB | 16s rRNase | ImmE6[28]
|
| Colicin E7 | A | BtuB | OmpF/TolQRAB | DNase | Im7[29]
|
| Colicin E8 | A | BtuB | OmpF/TolQRAB | DNase | Im8[30]
|
| Colicin E9 | A | BtuB | OmpF/TolQRAB | DNase | Im9[31]
|
| Colicin N | A | OmpF | OmpF/TolQRA | Pore-forming | Cni[32]
|
| Colicin S4 | A | OmpW | OmpF/TolQRAB | Pore-forming | Csi[33]
|
| Colicin K | A | Tsx | OmpF/TolQRAB | Pore-forming | ?
|
| Cloacin DF13 | A | lutA [34] | TolQRA [35] | 16s rRNase [36] |
|
| Colicin U | A | ? | OmpAF, TolQRAB | Pore-forming | Cui[37]
|
| Colicin 5 | B | Tsx | TolC/TonB, ExbBD | Pore-forming | Cfi[38]
|
| Colicin 6 | B | Tsx | TolC/TonB, ExbBD | Pore-forming | ?
|
| Colicin 7 | B | Tsx | TolC/TonB, ExbBD | Pore-forming | ?
|
| Colicin 8 | B | Tsx | TolC/TonB, ExbBD | Pore-forming | ?
|
| Colicin 9 | B | Tsx | TolC/TonB, ExbBD | Pore-forming | ?
|
| Colicin 10 | B | Tsx | TolC/TonB, ExbBD | Pore-forming | Cti[39]
|
| Colicin Ia | B | Cir | Cir/TonB, ExbBD | Pore-forming | Iia[40]
|
| Colicin Ib | B | Cir | Cir/TonB, ExbBD | Pore-forming | Imm
|
| Colicin B | B | FepA | ?/TonB, ExbBD | Pore-forming | Cbi[41]
|
| Colicin D | B | FepA | ?/TonB, ExbBD | tRNase | ImmD[42]
|
| Colicin M | B | FhuA | ?/TonB, ExbBD | Inhibition of PG synthesis | Cmi[43]
|
| Colicin V | B | Cir? [44] | TonB, ExbB | Disruption of membrane potential | Cvi[45]
|
| Colicin Js | B | CjrBC [46] | ExbBD, VirB [47] | ? | Cji [48]
|
| Colicin Y | ? | ? | ? | Pore-forming [49] | Cyi[50]
|
Table taken from [51] except where indicated.
SEE ALSO
Colicin Immunity Protein
- 1col – EcColA pore-forming domain
- 1tfk, 1tfo, 1v74 - EcColD catalytic domain + EcColD immunity protein
- 2i88 – EcColE1 channel-forming domain
- 2ysu – EcColE2 receptor-binding domain + BtuB
- 3u43 – EcColE2 nuclease domain + EcColE2 immunity protein
- 4v5k – EcColE3 cytotoxic domain (mutant) + Tt30S ribosome – Thermos thermophilus
- 2zld - EcColE3 cytotoxic domain + outer membrane protein F
- 1jch – EcColE3 + EcColE3 immunity protein
- 2b5u – EcColE3 (mutant) + EcColE3 immunity protein
- 1e44 – EcColE3 nuclease domain + EcColE3 immunity protein
- 1ujw – EcColE3 receptor-binding domain + BtuB
- 2djh, 3ao9 – EcColE5 C-terminal domain
- 3vj7 - EcColE5 C-terminal domain (mutant)
- 2a8k – EcColE5 catalytic domain
- 2dfx – EcColE5 C-terminal domain + EcColE5 immunity protein
- 2fhz – EcColE5 residues 74-180 + EcColE5 immunity protein
- 1unk - EcColE7
- 2axc - EcColE7 translocation domain
- 1m08, 3zfk - EcColE7 nuclease domain
- 3fbd, 1zns, 2ivh – EcColE7 (mutant) + DNA
- 1pt3 - EcColE7 nuclease domain + DNA
- 1mz8, 7cei - EcColE7 nuclease domain + EcColE7 immunity protein
- 3gjn - EcColE7 nuclease domain + EcColE9 immunity protein (mutant)
- 3gkl - EcColE7 nuclease domain (mutant) + EcColE9 immunity protein (mutant)
- 2jaz, 2jb0, 2jbg, 1znv - EcColE7 nuclease domain (mutant) + EcColE7 immunity protein
- 2erh, 1ujz - EcColE7 (mutant) + EcColE7 immunity protein (mutant)
- 1fsj - EcColE9 DNase domain
- 1v13 - EcColE9 DNase domain (mutant)
- 1v14, 1v15 - EcColE9 DNase domain (mutant) + DNA
- 2wpt – EcColE9 (mutant) + EcColE2 immunity protein
- 2k5x, 1emv, 1bxi – EcColE9 DNase domain + EcColE9 immunity protein
- 2vln, 2vlo, 2vlp, 2vlq - EcColE9 DNase domain (mutant) + EcColE9 immunity protein
- 2gze, 2gzg, 2gzi, 2gzj, 2gzf, 2gyk, 1fr2 - EcColE9 DNase domain + EcColE9 immunity protein (mutant)
- 2ivz - EcColE9 T domain + TolB
- 3o0e - EcColE9 fragment + outer membrane porin 1A
- 1cii - EcColIa
- 2hdi – EcColIA R domain + ColI receptor
- 1a87 – EcColN receptor-binding domain
}}
References
- ↑ Hands SL, Holland LE, Vankemmelbeke M, Fraser L, Macdonald CJ, Moore GR, James R, Penfold CN. Interactions of TolB with the translocation domain of colicin E9 require an extended TolB box. J Bacteriol. 2005 Oct;187(19):6733-41. PMID:16166536 doi:10.1128/JB.187.19.6733-6741.2005
- ↑ Cascales E, Buchanan SK, Duche D, Kleanthous C, Lloubes R, Postle K, Riley M, Slatin S, Cavard D. Colicin biology. Microbiol Mol Biol Rev. 2007 Mar;71(1):158-229. PMID:17347522 doi:10.1128/MMBR.00036-06
- ↑ Riley MA, Wertz JE. Bacteriocin diversity: ecological and evolutionary perspectives. Biochimie. 2002 May-Jun;84(5-6):357-64. PMID:12423779
- ↑ Czaran TL, Hoekstra RF, Pagie L. Chemical warfare between microbes promotes biodiversity. Proc Natl Acad Sci U S A. 2002 Jan 22;99(2):786-90. Epub 2002 Jan 15. PMID:11792831 doi:10.1073/pnas.012399899
- ↑ Kerr B, Riley MA, Feldman MW, Bohannan BJ. Local dispersal promotes biodiversity in a real-life game of rock-paper-scissors. Nature. 2002 Jul 11;418(6894):171-4. PMID:12110887 doi:10.1038/nature00823
- ↑ Wallis R, Moore GR, James R, Kleanthous C. Protein-protein interactions in colicin E9 DNase-immunity protein complexes. 1. Diffusion-controlled association and femtomolar binding for the cognate complex. Biochemistry. 1995 Oct 24;34(42):13743-50. PMID:7577966
- ↑ Ghosh P, Mel SF, Stroud RM. The domain structure of the ion channel-forming protein colicin Ia. Nat Struct Biol. 1994 Sep;1(9):597-604. PMID:7543362
- ↑ Pugsley AP. Genetic analysis of ColN plasmid determinants for colicin production, release, and immunity. J Bacteriol. 1984 May;158(2):523-9. PMID:6327609
- ↑ Penfold CN, Healy B, Housden NG, Boetzel R, Vankemmelbeke M, Moore GR, Kleanthous C, James R. Flexibility in the receptor-binding domain of the enzymatic colicin E9 is required for toxicity against Escherichia coli cells. J Bacteriol. 2004 Jul;186(14):4520-7. PMID:15231784 doi:10.1128/JB.186.14.4520-4527.2004
- ↑ Braun V, Patzer SI, Hantke K. Ton-dependent colicins and microcins: modular design and evolution. Biochimie. 2002 May-Jun;84(5-6):365-80. PMID:12423780
- ↑ Mock M, Pugsley AP. The BtuB group col plasmids and homology between the colicins they encode. J Bacteriol. 1982 Jun;150(3):1069-76. PMID:6281233
- ↑ Braun V, Patzer SI, Hantke K. Ton-dependent colicins and microcins: modular design and evolution. Biochimie. 2002 May-Jun;84(5-6):365-80. PMID:12423780
- ↑ Cascales E, Buchanan SK, Duche D, Kleanthous C, Lloubes R, Postle K, Riley M, Slatin S, Cavard D. Colicin biology. Microbiol Mol Biol Rev. 2007 Mar;71(1):158-229. PMID:17347522 doi:10.1128/MMBR.00036-06
- ↑ Bainbridge G, Armstrong GA, Dover LG, Whelan KF, Lakey JH. Displacement of OmpF loop 3 is not required for the membrane translocation of colicins N and A in vivo. FEBS Lett. 1998 Aug 7;432(3):117-22. PMID:9720908
- ↑ Benedetti H, Lloubes R, Lazdunski C, Letellier L. Colicin A unfolds during its translocation in Escherichia coli cells and spans the whole cell envelope when its pore has formed. EMBO J. 1992 Feb;11(2):441-7. PMID:1537329
- ↑ Cowan SW, Schirmer T, Rummel G, Steiert M, Ghosh R, Pauptit RA, Jansonius JN, Rosenbusch JP. Crystal structures explain functional properties of two E. coli porins. Nature. 1992 Aug 27;358(6389):727-33. PMID:1380671 doi:http://dx.doi.org/10.1038/358727a0
- ↑ Buchanan SK, Lukacik P, Grizot S, Ghirlando R, Ali MM, Barnard TJ, Jakes KS, Kienker PK, Esser L. Structure of colicin I receptor bound to the R-domain of colicin Ia: implications for protein import. EMBO J. 2007 May 16;26(10):2594-604. Epub 2007 Apr 26. PMID:17464289
- ↑ Mosbahi K, Walker D, Lea E, Moore GR, James R, Kleanthous C. Destabilization of the colicin E9 Endonuclease domain by interaction with negatively charged phospholipids: implications for colicin translocation into bacteria. J Biol Chem. 2004 May 21;279(21):22145-51. Epub 2004 Mar 23. PMID:15044477 doi:10.1074/jbc.M400402200
- ↑ Nomura M. Colicins and related bacteriocins. Annu Rev Microbiol. 1967;21:257-84. PMID:4860260 doi:http://dx.doi.org/10.1146/annurev.mi.21.100167.001353
- ↑ Cutler SA, Lonergan SM, Cornick N, Johnson AK, Stahl CH. Dietary inclusion of colicin e1 is effective in preventing postweaning diarrhea caused by F18-positive Escherichia coli in pigs. Antimicrob Agents Chemother. 2007 Nov;51(11):3830-5. Epub 2007 Aug 27. PMID:17724148 doi:10.1128/AAC.00360-07
- ↑ Gillor O, Kirkup BC, Riley MA. Colicins and microcins: the next generation antimicrobials. Adv Appl Microbiol. 2004;54:129-46. PMID:15251279 doi:10.1016/S0065-2164(04)54005-4
- ↑ Nardi A, Corda Y, Baty D, Duche D. Colicin A immunity protein interacts with the hydrophobic helical hairpin of the colicin A channel domain in the Escherichia coli inner membrane. J Bacteriol. 2001 Nov;183(22):6721-5. PMID:11673448 doi:10.1128/JB.183.22.6721-6725.2001
- ↑ Song HY, Cramer WA. Membrane topography of ColE1 gene products: the immunity protein. J Bacteriol. 1991 May;173(9):2935-43. PMID:1708384
- ↑ Duche D, Issouf M, Lloubes R. Immunity protein protects colicin E2 from OmpT protease. J Biochem. 2009 Jan;145(1):95-101. Epub 2008 Nov 5. PMID:18990718 doi:10.1093/jb/mvn149
- ↑ Jakes K, Zinder ND, Boon T. Purification and properties of colicin E3 immunity protein. J Biol Chem. 1974 Jan 25;249(2):438-44. PMID:4588566
- ↑ Carr S, Walker D, James R, Kleanthous C, Hemmings AM. Crystallization of the cytotoxic domain of a ribosome-inactivating colicin in complex with its immunity protein. Acta Crystallogr D Biol Crystallogr. 2000 Dec;56(Pt 12):1630-3. PMID:11092930
- ↑ Lau PC, Condie JA. Nucleotide sequences from the colicin E5, E6 and E9 operons: presence of a degenerate transposon-like structure in the ColE9-J plasmid. Mol Gen Genet. 1989 Jun;217(2-3):269-77. PMID:2549375
- ↑ Akutsu A, Masaki H, Ohta T. Molecular structure and immunity specificity of colicin E6, an evolutionary intermediate between E-group colicins and cloacin DF13. J Bacteriol. 1989 Dec;171(12):6430-6. PMID:2687234
- ↑ Dennis CA, Videler H, Pauptit RA, Wallis R, James R, Moore GR, Kleanthous C. A structural comparison of the colicin immunity proteins Im7 and Im9 gives new insights into the molecular determinants of immunity-protein specificity. Biochem J. 1998 Jul 1;333 ( Pt 1):183-91. PMID:9639578
- ↑ Keeble AH, Kleanthous C. The kinetic basis for dual recognition in colicin endonuclease-immunity protein complexes. J Mol Biol. 2005 Sep 23;352(3):656-71. PMID:16109424 doi:10.1016/j.jmb.2005.07.035
- ↑ Osborne MJ, Breeze AL, Lian LY, Reilly A, James R, Kleanthous C, Moore GR. Three-dimensional solution structure and 13C nuclear magnetic resonance assignments of the colicin E9 immunity protein Im9. Biochemistry. 1996 Jul 23;35(29):9505-12. PMID:8755730 doi:10.1021/bi960401k
- ↑ Geli V, Baty D, Pattus F, Lazdunski C. Topology and function of the integral membrane protein conferring immunity to colicin A. Mol Microbiol. 1989 May;3(5):679-87. PMID:2668695
- ↑ Pilsl H, Smajs D, Braun V. Characterization of colicin S4 and its receptor, OmpW, a minor protein of the Escherichia coli outer membrane. J Bacteriol. 1999 Jun;181(11):3578-81. PMID:10348872
- ↑ Wooldridge KG, Williams PH. Sensitivity of Escherichia coli to cloacin DF13 involves the major outer membrane protein OmpF. J Bacteriol. 1991 Apr;173(8):2420-4. PMID:2013565
- ↑ Thomas JA, Valvano MA. Role of tol genes in cloacin DF13 susceptibility of Escherichia coli K-12 strains expressing the cloacin DF13-aerobactin receptor IutA. J Bacteriol. 1993 Jan;175(2):548-52. PMID:8419302
- ↑ Baan RA, Duijfjes JJ, van Leerdam E, van Knippenberg PH, Bosch L. Specific in situ cleavage of 16S ribosomal RNA of Escherichia coli interferes with the function of initiation factor IF-1. Proc Natl Acad Sci U S A. 1976 Mar;73(3):702-6. PMID:768982
- ↑ Smajs D, Pilsl H, Braun V. Colicin U, a novel colicin produced by Shigella boydii. J Bacteriol. 1997 Aug;179(15):4919-28. PMID:9244283
- ↑ Pilsl H, Braun V. Evidence that the immunity protein inactivates colicin 5 immediately prior to the formation of the transmembrane channel. J Bacteriol. 1995 Dec;177(23):6966-72. PMID:7592492
- ↑ Pilsl H, Braun V. Novel colicin 10: assignment of four domains to TonB- and TolC-dependent uptake via the Tsx receptor and to pore formation. Mol Microbiol. 1995 Apr;16(1):57-67. PMID:7651137
- ↑ Weaver CA, Redborg AH, Konisky J. Plasmid-determined immunity of Escherichia coli K-12 to colicin Ia Is mediated by a plasmid-encoded membrane protein. J Bacteriol. 1981 Dec;148(3):817-28. PMID:6273383
- ↑ Schramm E, Olschlager T, Troger W, Braun V. Sequence, expression and localization of the immunity protein for colicin B. Mol Gen Genet. 1988 Jan;211(1):176-82. PMID:2830463
- ↑ Graille M, Mora L, Buckingham RH, van Tilbeurgh H, de Zamaroczy M. Structural inhibition of the colicin D tRNase by the tRNA-mimicking immunity protein. EMBO J. 2004 Apr 7;23(7):1474-82. Epub 2004 Mar 11. PMID:15014439 doi:10.1038/sj.emboj.7600162
- ↑ Olschlager T, Schramm E, Braun V. Cloning and expression of the activity and immunity genes of colicins B and M on ColBM plasmids. Mol Gen Genet. 1984;196(3):482-7. PMID:6094976
- ↑ Waters VL, Crosa JH. Colicin V virulence plasmids. Microbiol Rev. 1991 Sep;55(3):437-50. PMID:1943995
- ↑ Frick KK, Quackenbush RL, Konisky J. Cloning of immunity and structural genes for colicin V. J Bacteriol. 1981 Nov;148(2):498-507. PMID:6271732
- ↑ Braun V, Patzer SI, Hantke K. Ton-dependent colicins and microcins: modular design and evolution. Biochimie. 2002 May-Jun;84(5-6):365-80. PMID:12423780
- ↑ Smajs D, Weinstock GM. The iron- and temperature-regulated cjrBC genes of Shigella and enteroinvasive Escherichia coli strains code for colicin Js uptake. J Bacteriol. 2001 Jul;183(13):3958-66. PMID:11395459 doi:10.1128/JB.183.13.3958-3966.2001
- ↑ Smajs D, Weinstock GM. Genetic organization of plasmid ColJs, encoding colicin Js activity, immunity, and release genes. J Bacteriol. 2001 Jul;183(13):3949-57. PMID:11395458 doi:10.1128/JB.183.13.3949-3957.2001
- ↑ Riley MA, Cadavid L, Collett MS, Neely MN, Adams MD, Phillips CM, Neel JV, Friedman D. The newly characterized colicin Y provides evidence of positive selection in pore-former colicin diversification. Microbiology. 2000 Jul;146 ( Pt 7):1671-7. PMID:10878131
- ↑ Smajs D, Matejkova P, Weinstock GM. Recognition of pore-forming colicin Y by its cognate immunity protein. FEMS Microbiol Lett. 2006 May;258(1):108-13. PMID:16630264 doi:10.1111/j.1574-6968.2006.00201.x
- ↑ Kleanthous C. Swimming against the tide: progress and challenges in our understanding of colicin translocation. Nat Rev Microbiol. 2010 Dec;8(12):843-8. Epub 2010 Nov 9. PMID:21060316 doi:10.1038/nrmicro2454
|



