This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
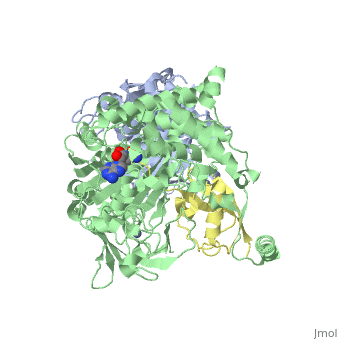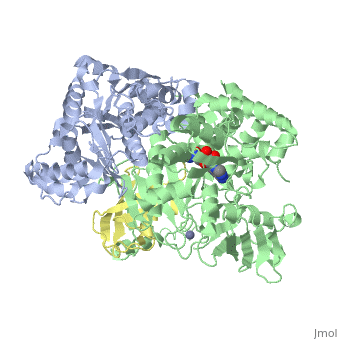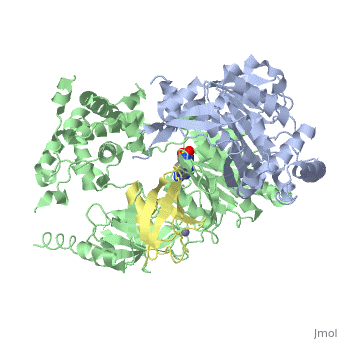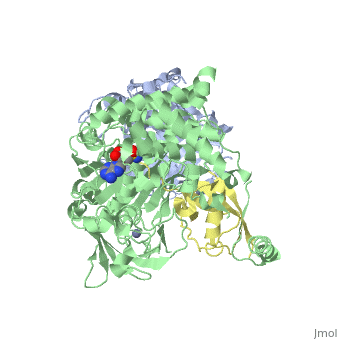
Human SUMO E1 complex
From Proteopedia
| Line 1: | Line 1: | ||
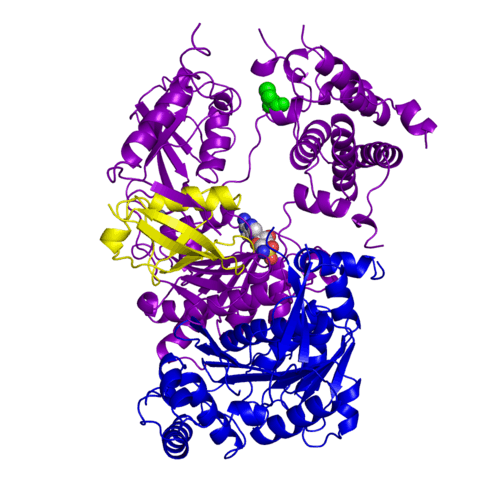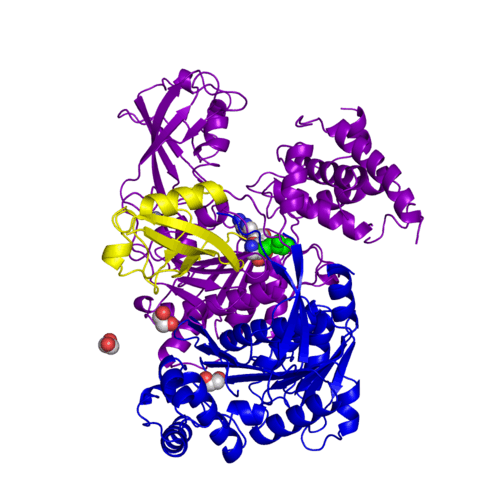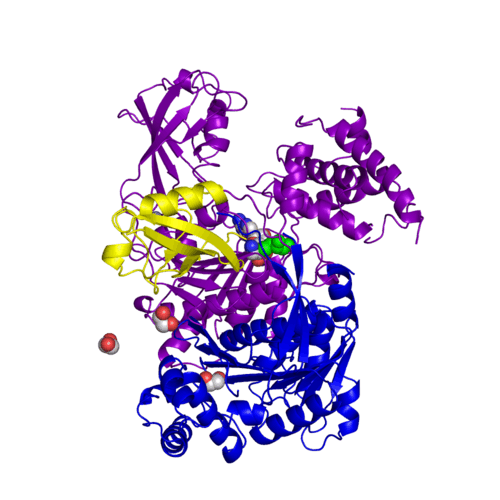
| - | <StructureSection load='3kyc' size= | + | <StructureSection load='3kyc' size="400" color="" spin="on" Scene= caption='Human SUMO-activating enzyme subunits 1 & 2 complex with SUMO-1, deoxy-sulfamoylamino-adenosine and Zn+2 ion [[3kyc]] > |
Ubiquitin (Ub) and ubiquitin-like (Ubl) proteins attached to their target proteins and modulating the activities of those targets in various ways. Three types of evolutionarily conserved enzymes — E1 activating enzymes, E2 conjugating enzymes and E3 ligase enzymes — act sequentially through parallel yet distinct pathways to conjugate ubiquitin and Ubl proteins, such as SUMO and NEDD8, to their targets. The E1 enzyme uses the <scene name='3kyc/Cv/3'>adenosine triphosphate (ATP)</scene> and magnesium to adenylate the C-terminal Ub/Ubl glycine, releasing pyrophosphate and resulting in <scene name='3kyc/Cv/8'>adenosine monophosphate (AMP)</scene>. A non-hydrolysable <scene name='3kyc/Cv/4'>mimic of the acyl adenylate intermediate (AMSN)</scene> and <scene name='3kyc/Cv/5'>mimic of the tetrahedral intermediate (AVSN)</scene> were constructed. In both these compounds the atom of <font color='orange'><b>phosphorus</b></font> is replaced by sulfur (colored <span style="color:yellow;background-color:black;font-weight:bold;">yellow</span>). | Ubiquitin (Ub) and ubiquitin-like (Ubl) proteins attached to their target proteins and modulating the activities of those targets in various ways. Three types of evolutionarily conserved enzymes — E1 activating enzymes, E2 conjugating enzymes and E3 ligase enzymes — act sequentially through parallel yet distinct pathways to conjugate ubiquitin and Ubl proteins, such as SUMO and NEDD8, to their targets. The E1 enzyme uses the <scene name='3kyc/Cv/3'>adenosine triphosphate (ATP)</scene> and magnesium to adenylate the C-terminal Ub/Ubl glycine, releasing pyrophosphate and resulting in <scene name='3kyc/Cv/8'>adenosine monophosphate (AMP)</scene>. A non-hydrolysable <scene name='3kyc/Cv/4'>mimic of the acyl adenylate intermediate (AMSN)</scene> and <scene name='3kyc/Cv/5'>mimic of the tetrahedral intermediate (AVSN)</scene> were constructed. In both these compounds the atom of <font color='orange'><b>phosphorus</b></font> is replaced by sulfur (colored <span style="color:yellow;background-color:black;font-weight:bold;">yellow</span>). | ||
| - | |||
The <scene name='3kyc/Al/2'>structural alignment</scene> of the crystal structures for human SUMO E1 in complex with SUMO adenylate (AMSN) and tetrahedral intermediate (AVSN) analogues revealed opened conformation (<font color='orange'><b>SUMO1 in orange</b></font>, <font color='blue'><b>SAE1 colored in blue</b></font>, and <font color='darkviolet'><b>other domains in darkviolet</b></font>) and closed conformation (<span style="color:yellow;background-color:black;font-weight:bold;">SUMO1 in yellow</span>, <font color='cyan'><b>SAE1 colored in cyan</b></font>, and <font color='magenta'><b>other domains in magenta</b></font>), respectively. In the <scene name='3kyc/Al/7'>open conformation</scene> ([[3kyc]]) the distance between Cys domain (including Cys173) and mimic of the acyl adenylate intermediate AMSN is very long, while in the <scene name='3kyc/Al/6'>closed conformation</scene> ([[3kyd]]), the catalytic Cys173 is posioned near AVSN and SUMO1, so the overall structure revealed dramatic rearrangement. This large conformational change forms the <scene name='3kyc/Al/8'>E1~SUMO1-AVSN tetrahedral intermediate analogue</scene>. | The <scene name='3kyc/Al/2'>structural alignment</scene> of the crystal structures for human SUMO E1 in complex with SUMO adenylate (AMSN) and tetrahedral intermediate (AVSN) analogues revealed opened conformation (<font color='orange'><b>SUMO1 in orange</b></font>, <font color='blue'><b>SAE1 colored in blue</b></font>, and <font color='darkviolet'><b>other domains in darkviolet</b></font>) and closed conformation (<span style="color:yellow;background-color:black;font-weight:bold;">SUMO1 in yellow</span>, <font color='cyan'><b>SAE1 colored in cyan</b></font>, and <font color='magenta'><b>other domains in magenta</b></font>), respectively. In the <scene name='3kyc/Al/7'>open conformation</scene> ([[3kyc]]) the distance between Cys domain (including Cys173) and mimic of the acyl adenylate intermediate AMSN is very long, while in the <scene name='3kyc/Al/6'>closed conformation</scene> ([[3kyd]]), the catalytic Cys173 is posioned near AVSN and SUMO1, so the overall structure revealed dramatic rearrangement. This large conformational change forms the <scene name='3kyc/Al/8'>E1~SUMO1-AVSN tetrahedral intermediate analogue</scene>. | ||
Current revision
| |||||||||||
For better understanding of the difference between these two conformations you can see this morph (generated by using POLYVIEW-3D: http://polyview.cchmc.org/polyview3d.html; reload/refresh this page to restart this movie). Of note, in contrast to the previous figure, the same domains of these two structures (3kyc and 3kyd) are colored in the same colors (SUMO1 in yellow, SAE1 colored in blue and other domains in darkviolet). The catalytic Cys173 is shown in the spacefill representation and colored green, AMSN (or AVSN) are shown in the spacefill representation and colored in CPK colors.
3D structures of SUMO
Reference
- Olsen SK, Capili AD, Lu X, Tan DS, Lima CD. Active site remodelling accompanies thioester bond formation in the SUMO E1. Nature. 2010 Feb 18;463(7283):906-12. PMID:20164921 doi:10.1038/nature08765
Categories: Topic Page | Homo sapiens | Lima, C D. | Acetylation | Adenylation | Atp-binding | Cytoplasm | E1 | Inhibitor | Isopeptide bond | Ligase | Membrane | Nucleotide-binding | Nucleus | Phosphoprotein | Polymorphism | Sumo | Tetrahedral intermediate | Thioester | Ubiquitin | Ubl conjugation pathway