User:Ashley Crotteau/Sandbox1
From Proteopedia
(Difference between revisions)
| Line 26: | Line 26: | ||
| + | == Function == | ||
| + | [[Image:KMT Mechanism .png|500 px|right|thumb|Figure 3: Histone Methylation Mechanism]] | ||
| + | ===Mechanism=== | ||
| + | ===Conserved Residues=== | ||
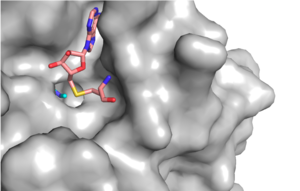
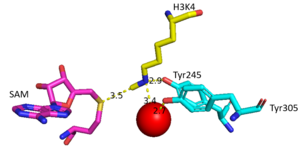
| - | == | + | The conserved regions of KMT has been found to be vital for function. Multiple studies have found that a mutation to any of the conserved tyrosine residues continues to monomethylate SAM, while it also creates di-methylation and tri- methylation of SAM.<ref name="Del Rizzo" /> In a recent study, Y245A and Y305F were created through site-directed mutagenesis.<ref name="Del Rizzo" /> Due to size, Ala245 was found to create a larger opening of the channel than tyrosine, which allowed for further methylation of SAM. <ref name="Del Rizzo" /> However, Y305F also showed the same characteristics of di- and tri-methylation, most likely due to a decrease of tyrosine residue interaction with water.<ref name="Del Rizzo" /> As there are four invariant conserved tyrosine residues (Tyr305, Tyr245, Tyr335, Tyr337) in the active site, this finding indicates that the function of KMT is dependent on the presence of tyrosine residues in the active site. <ref name="Del Rizzo" /> |
| - | + | ||
== Relevance == | == Relevance == | ||
| - | === | + | ===Renal Fibrosis=== |
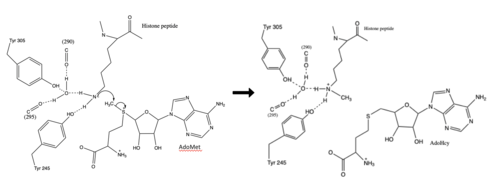
| + | [[Image:KMT Mechanism .png|500 px|right|thumb|Figure 4: Structural differences between SAM and Sinefungin]] | ||
| + | The inhibition of KMT SET 7/9 has been found to enhance renal fibrosis.<ref name="Sun" /> H3K4 methylation activates the transcription of fibrotic genes, and the suppression of the H3K4 methylation was found to enhance renal fibrosis in a mouse model.<ref name="Sun" /> Sinefungin, a competitive methyltransferase inhibitor, binds to KMT to inhibit SAM.<ref name="Sun" /> SAM and Sinefungin have similar structures (Figure 4), differing only with the removal of a sulfide and the replacement of a methyl to an amine. The amine replaces the methyl donor for the reaction of KMT, inhibiting the reaction and preventing methylation. Without the methylation of H3K4, the transcription of fibrotic genes is deactivated, leading to renal fibrosis.<ref name="Sun" /> | ||
| + | |||
| + | ===Cancer=== | ||
| + | |||
| Line 45: | Line 53: | ||
<ref name="Huang">PMID: 9632640 </ref> | <ref name="Huang">PMID: 9632640 </ref> | ||
<ref name="Wikipedia"> https://en.wikipedia.org/wiki/SET_domain#Structure </ref> | <ref name="Wikipedia"> https://en.wikipedia.org/wiki/SET_domain#Structure </ref> | ||
| + | <ref name="Del Rizzo">PMID: 20675860 </ref> | ||
| + | <ref name="Sun">PMID: 20930066 </ref> | ||
<references/> | <references/> | ||
Revision as of 18:29, 5 April 2019
H. sapiens Lysine Methyltransferase, SET 7/9
| |||||||||||
References
[3] [5] [1] [2] [4] [8] [6] [7]
- ↑ 1.0 1.1 1.2 1.3 1.4 1.5 1.6 Schubert HL, Blumenthal RM, Cheng X. Many paths to methyltransfer: a chronicle of convergence. Trends Biochem Sci. 2003 Jun;28(6):329-35. PMID:12826405
- ↑ 2.0 2.1 2.2 2.3 2.4 2.5 Yeates TO. Structures of SET domain proteins: protein lysine methyltransferases make their mark. Cell. 2002 Oct 4;111(1):5-7. PMID:12372294
- ↑ 3.0 3.1 3.2 3.3 3.4 3.5 3.6 3.7 3.8 Xiao B, Jing C, Wilson JR, Walker PA, Vasisht N, Kelly G, Howell S, Taylor IA, Blackburn GM, Gamblin SJ. Structure and catalytic mechanism of the human histone methyltransferase SET7/9. Nature. 2003 Feb 6;421(6923):652-6. Epub 2003 Jan 22. PMID:12540855 doi:10.1038/nature01378
- ↑ 4.0 4.1 Huang S, Shao G, Liu L. The PR domain of the Rb-binding zinc finger protein RIZ1 is a protein binding interface and is related to the SET domain functioning in chromatin-mediated gene expression. J Biol Chem. 1998 Jun 26;273(26):15933-9. PMID:9632640
- ↑ 5.0 5.1 doi: https://dx.doi.org/10.1016/C2014-0-02189-2
- ↑ 6.0 6.1 6.2 6.3 6.4 6.5 Del Rizzo PA, Couture JF, Dirk LM, Strunk BS, Roiko MS, Brunzelle JS, Houtz RL, Trievel RC. SET7/9 catalytic mutants reveal the role of active site water molecules in lysine multiple methylation. J Biol Chem. 2010 Oct 8;285(41):31849-58. Epub 2010 Aug 1. PMID:20675860 doi:http://dx.doi.org/10.1074/jbc.M110.114587
- ↑ 7.0 7.1 7.2 7.3 7.4 Sun G, Reddy MA, Yuan H, Lanting L, Kato M, Natarajan R. Epigenetic histone methylation modulates fibrotic gene expression. J Am Soc Nephrol. 2010 Dec;21(12):2069-80. doi: 10.1681/ASN.2010060633. Epub 2010, Oct 7. PMID:20930066 doi:http://dx.doi.org/10.1681/ASN.2010060633
- ↑ https://en.wikipedia.org/wiki/SET_domain#Structure
Student Contributors
Ashley Crotteau
Parker Hiday
Lauren Allman