This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Sandbox Reserved 1692
From Proteopedia
(Difference between revisions)
| Line 11: | Line 11: | ||
== Important amino acids== | == Important amino acids== | ||
Amino Acids <scene name='89/892735/Amino_acids2/2'>His85, Asp106, Tyr150, Ser170, Tyr202, Arg220, Pro223, Ala225, and Arg331</scene> provide important interactions for binding in the active site. The <scene name='89/892735/His85_and_his105/1'>His105 side chain forms a hydrogen bond with His85</scene> side chain, providing stabilizing assistance. <ref name="Kondo">PMID:34303708</ref>. | Amino Acids <scene name='89/892735/Amino_acids2/2'>His85, Asp106, Tyr150, Ser170, Tyr202, Arg220, Pro223, Ala225, and Arg331</scene> provide important interactions for binding in the active site. The <scene name='89/892735/His85_and_his105/1'>His105 side chain forms a hydrogen bond with His85</scene> side chain, providing stabilizing assistance. <ref name="Kondo">PMID:34303708</ref>. | ||
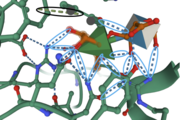
| - | Amino Acid Residues <scene name='89/892735/Tyr150_and_his85/1'>Tyr150 and His85</scene> form a hydrogen bond with the O-1 atom of Rha, suggesting these residues play a catalytic role for the elimination reaction. [[Image:IntermolforcesBIOCHEM.png|thumb]] | + | Amino Acid Residues <scene name='89/892735/Tyr150_and_his85/1'>Tyr150 and His85</scene> form a hydrogen bond with the O-1 atom of Rha, suggesting these residues play a catalytic role for the elimination reaction. <scene name='89/892735/O2o3hbonds/1'>Tyr26, Asn275, Gln276, and Arg331</scene> form hydrogen bonds with O-2 and O-3 atoms of Rha.[[Image:IntermolforcesBIOCHEM.png|thumb]] |
The image to the right shows important amino acids in the active site. Hydrogen bonding and pi-stacking interactions are indicated by the blue and black circles, respectively. | The image to the right shows important amino acids in the active site. Hydrogen bonding and pi-stacking interactions are indicated by the blue and black circles, respectively. | ||
== Structural highlights == | == Structural highlights == | ||
Revision as of 17:35, 8 December 2021
| This Sandbox is Reserved from 10/01/2021 through 01/01//2022 for use in Biochemistry taught by Bonnie Hall at Grand View University, Des Moines, USA. This reservation includes Sandbox Reserved 1690 through Sandbox Reserved 1699. |
To get started:
More help: Help:Editing |
Structure and Function of FoRham1
| |||||||||||
References
- ↑ 1.0 1.1 1.2 Kondo T, Kichijo M, Maruta A, Nakaya M, Takenaka S, Arakawa T, Fushinobu S, Sakamoto T. Structural and functional analysis of gum arabic l-rhamnose-alpha-1,4-d-glucuronate lyase establishes a novel polysaccharide lyase family. J Biol Chem. 2021 Jul 22:101001. doi: 10.1016/j.jbc.2021.101001. PMID:34303708 doi:http://dx.doi.org/10.1016/j.jbc.2021.101001