We apologize for Proteopedia being slow to respond. For the past two years, a new implementation of Proteopedia has been being built. Soon, it will replace this 18-year old system. All existing content will be moved to the new system at a date that will be announced here.
3atg
From Proteopedia
(Difference between revisions)
| Line 3: | Line 3: | ||
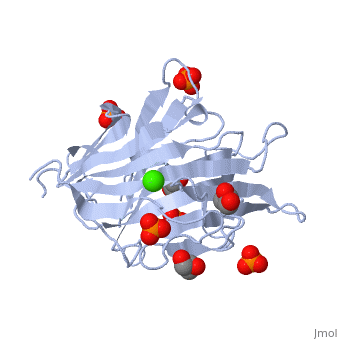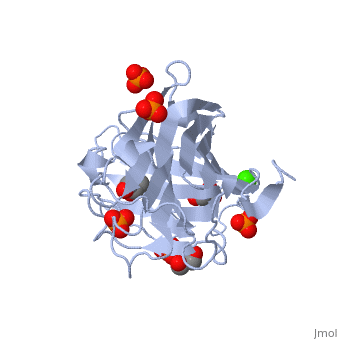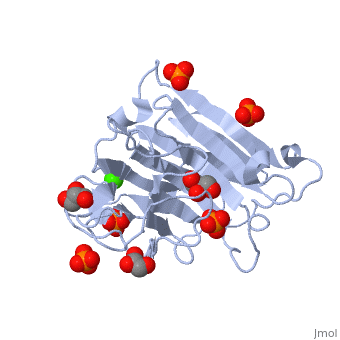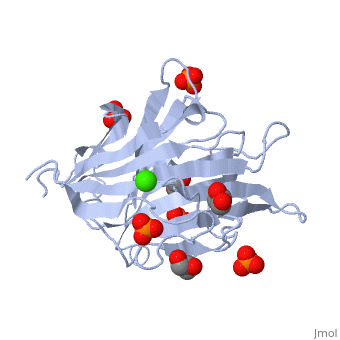
<StructureSection load='3atg' size='340' side='right'caption='[[3atg]], [[Resolution|resolution]] 1.66Å' scene=''> | <StructureSection load='3atg' size='340' side='right'caption='[[3atg]], [[Resolution|resolution]] 1.66Å' scene=''> | ||
== Structural highlights == | == Structural highlights == | ||
| - | <table><tr><td colspan='2'>[[3atg]] is a 1 chain structure with sequence from [ | + | <table><tr><td colspan='2'>[[3atg]] is a 1 chain structure with sequence from [https://en.wikipedia.org/wiki/Cellulosimicrobium_cellulans Cellulosimicrobium cellulans]. Full crystallographic information is available from [http://oca.weizmann.ac.il/oca-bin/ocashort?id=3ATG OCA]. For a <b>guided tour on the structure components</b> use [https://proteopedia.org/fgij/fg.htm?mol=3ATG FirstGlance]. <br> |
| - | </td></tr><tr id='ligand'><td class="sblockLbl"><b>[[Ligand|Ligands:]]</b></td><td class="sblockDat"><scene name='pdbligand=CA:CALCIUM+ION'>CA</scene>, <scene name='pdbligand=GOL:GLYCEROL'>GOL</scene>, <scene name='pdbligand=PO4:PHOSPHATE+ION'>PO4</scene></td></tr> | + | </td></tr><tr id='ligand'><td class="sblockLbl"><b>[[Ligand|Ligands:]]</b></td><td class="sblockDat" id="ligandDat"><scene name='pdbligand=CA:CALCIUM+ION'>CA</scene>, <scene name='pdbligand=GOL:GLYCEROL'>GOL</scene>, <scene name='pdbligand=PO4:PHOSPHATE+ION'>PO4</scene></td></tr> |
| - | <tr id='activity'><td class="sblockLbl"><b>Activity:</b></td><td class="sblockDat"><span class='plainlinks'>[ | + | <tr id='activity'><td class="sblockLbl"><b>Activity:</b></td><td class="sblockDat"><span class='plainlinks'>[https://en.wikipedia.org/wiki/Glucan_endo-1,3-beta-D-glucosidase Glucan endo-1,3-beta-D-glucosidase], with EC number [https://www.brenda-enzymes.info/php/result_flat.php4?ecno=3.2.1.39 3.2.1.39] </span></td></tr> |
| - | <tr id='resources'><td class="sblockLbl"><b>Resources:</b></td><td class="sblockDat"><span class='plainlinks'>[ | + | <tr id='resources'><td class="sblockLbl"><b>Resources:</b></td><td class="sblockDat"><span class='plainlinks'>[https://proteopedia.org/fgij/fg.htm?mol=3atg FirstGlance], [http://oca.weizmann.ac.il/oca-bin/ocaids?id=3atg OCA], [https://pdbe.org/3atg PDBe], [https://www.rcsb.org/pdb/explore.do?structureId=3atg RCSB], [https://www.ebi.ac.uk/pdbsum/3atg PDBsum], [https://prosat.h-its.org/prosat/prosatexe?pdbcode=3atg ProSAT]</span></td></tr> |
</table> | </table> | ||
<div style="background-color:#fffaf0;"> | <div style="background-color:#fffaf0;"> | ||
Revision as of 15:03, 29 December 2021
endo-1,3-beta-glucanase from Cellulosimicrobium cellulans
| |||||||||||