Introduction
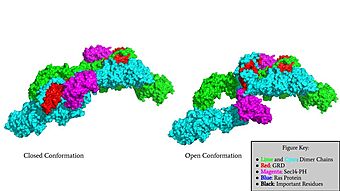
Surface Rendering of Neurofibromin in its Open and Closed Conformation
Neurofibromin is encoded by NF1 gene, located on chromosome 17. Neurofibromin functions as a tumor suppressor through its association with the protein Ras. The molecular structure of Neurofibromin has been determined by Cryo-Electron Microscopy. The structure of neurofibromin isoform 2 revealed different functional states for the Neurofibromin protein.[1] Mutations in Neurofibromin areassociated with diseases such as Plexiform Neurofibromas.
Function
Neurofibromin functions as a tumor suppressor protein.[2] Its job is to prevent cell growth by turning off another protein known as Ras which in its active state, stimulates cell growth and division. Ras is a GTPase membrane protein that can only interact with Neurofibromin, a cytoplasmic protein, in the open conformation of Neurofibromin. As Neurofibromin is a cytoplasmic protein, it is brought to the membrane to associate with Ras via another protein known as SPRED1. Neurofibromin can interact with SPRED1 in both the open and closed conformations however, it can only associate with Ras when it is in its open conformation. The interaction between Neurofibromin and Ras occurs via an Arginine finger (R1276) present in the GRD domain of Neurofibromin which is critical for Ras binding. R1276 is only accessible for binding when the GRD and Sec14-PH domains are rotated into the open conformation and there is no steric hindrance from the surrounding dimer chains. When R1276 is able to associate with Ras, Neurofibromin downregulates the Ras signaling pathway by hydrolyzing the GTP associated with Ras to GDP, effectively making it inactive and inhibiting cell growth and division.
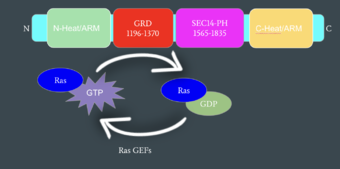
Mechanism of Ras Regulation by Neurofibromin
Structure
Neurofibromin is a made up of two identical chains. Neurofibromin has two conformations, open and closed. Shifting between these controls neurofibromins ability to associate with [hhttps://en.wikipedia.org/wiki/Ras_GTPase Ras](6ob3) and perform its function of Ras regulation. The transformation between the overall closed and open conformations transitions it from an active to inactive state. There are two important domains involved in the transition between the open and closed conformations, the domain and the domain. Although neurofibromin is a homodimer with two identical protomers, only one protomer has its GRD and Sec14-PH domains rotated into the open conformation.
Closed Conformation (7PGR)
In the inactive, , the GRD and Sec14-PH domains are rotated so Ras cannot bind. In this conformation, the GRD and Sec14-PH are inaccessible and inactive. Neurofibromin is held in the inactive state by a consisting of residues Cys 1032, His 1558, and His 1576 that form a transition metal-binding site with zinc. The rigid organization of the keeps the GRD domain packed tightly on top of the Heat Arms in the Neurofibromin core. This tight compaction sterically occludes Neurofibromin from associating with Ras. In its active form, Ras and Neurofibromin will associate via an (Arg 1276). However, the steric hindrance from the Neurofibromin core in the closed conformation inhibits this association. Therefore, in the closed conformation, neurofibromin cannot catalyze GTP hydrolysis by Ras and Ras continues to signal for cell growth and proliferation.
Open Conformation (7PGT)
The other conformation that characterizes Neurofibromin is the . In the open conformation of Neurofibromin, the protein is considered active and is participating in its function of Ras regulation. This occurs because the transition metal-binding site with zinc no longer is able to form due to an increase in distance between the C1032, H1558 and H1576 residues that form the . One protomer in Neurofibromin has its GRD and Sec14-PH domains oriented in way that is almost reversed in position compared to the closed conformation. The GRD rotates -130° and the Sec14-PH domain rotates -90° in the transition between the closed and the open conformation. Due to this rotation, C1032 is now located too far away, approximately 30 Angstroms, from H1558 and H1576 which results in the loss of the metal-binding site. The lack of the transition metal-binding site allows the GRD to orient itself in such a way that it can associate with . The reason that Neurofibromin is only able to associate with Ras in the open conformation is due to one critical residue, the located at position 1276 in Neurofibromin. When Neurofibromin is in the open conformation, R1276 is able to because there is no steric hindrance from the Neurofibromin core.
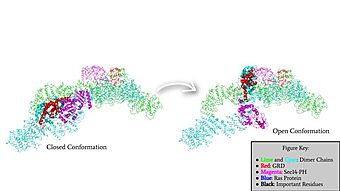
Rotation of the GRD and Sec14-PH domains from the closed conformation of neurofibromin to the open conformation of neurofibromin to allow Ras binding. The GRD rotates -130° and the Sec14-PH domain rotates -90°
Disease and Medical Relevance
Currently, there are over 1485 mutations of Neurofibromin that have been identified. Mutations in Neurofibromin can lead to life-threatening illnesses or conditions such as Neurofibromatosis Type I due to the inability of Neurofibromin to interact with Ras.[3] The role of Neurofibromin is to inhibit cellular proliferation via its Guanine triphosphatase-activating protein (GAP) activity; therefore, when there are mutations to the NF1 gene that prevent the interaction of Neufibromin with Ras, there is nothing stopping Ras from promoting cell growth. Uncontrolled cell growth can lead to tumors and a higher risk of cancer. Neurofibromatosis Type 1, the most well-known disease resulting from mutations in Neurofibromin, is characterized by cognitive impairment, soft, non-cancerous tumors on or under the skin known as neurofibromas, birthmarks called cafe-au-lait macules, clusters of freckles in unusual places, and problems with the bones, eyes and nervous system.
Neurofibromin is an essential protein and is involved mainly in the differentiation of neural crest-derived cells, mesenchymal cells, neural cells, melanocytes, and bone cells. As Neurofibromin is essential for embryonic development, mutations to the NF1 gene can result in psychological retardation resulting from Type I neurofibromatosis. Most of the 1485 mutations identified lead to a synthesis of truncated, non-functional protein and are a result of point mutations. Type I Neurofibromatosis is inherited in an autosomal dominant manner but about 50% of cases de novo ones. [4]
Mutations of the gene NF1 can lead to major structural and functional changes that are dangerous for the patient. NF1 mutation(s) can result in the complete loss of function and the inability to interact with the Ras protein. If interaction with Ras cannot occur, no conversion of GTP to GDP occurs and, therefore, no inactivation of Ras. Consequently, there is nothing limiting Ras from promoting cell growth. Uncontrolled cell growth leads to tumors and puts patients at a much higher risk for cancer. Mutations in the NF1 gene have been found in sporadic cancers such as glioblastoma, neuroblastoma, lung cancer, ovarian cancer, and breast cancer. Additionally, NF1 is one of the top ten genes that are most mutated in tumors of the lung, breast, ovary, pancreas, and prostate. Researchers are still unsure as to whether biallelic loss of NF1 is common or if it is only a hemizygous loss of the gene that contributes to the growth progression of certain sporadic tumors. It is possible that the order of mutations affects the grade of a tumor (for certain types of cells) and thus explains why NF1 patients are not predisposed to certain types of sporadic tumors.[5]
Student Contributors
- Hannah Luchinski
- Sophie Mullinix



