User:George G. Papadeas/Sandbox VKOR
From Proteopedia
(Difference between revisions)
| Line 12: | Line 12: | ||
== Structural Highlights== | == Structural Highlights== | ||
| + | ===Structural Overview=== | ||
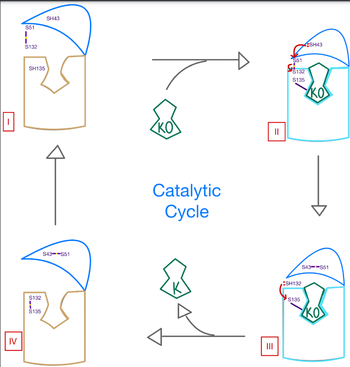
| + | VKOR has many key components of its structure that allow it to maintain proper functionality and catalytic abilities. The VKOR active site allows for specific substrate binding via many highly conserved residues that can recognize the target substrates. It works in conjunction with the cap domain, which is a helical component of the VKOR that facilitates the conformation from the open to closed conformation of the enzyme once the substrate binds. Interactions between this domain, the active site, and the bound protein are critical to achieve full activation of Vitamin K. Another important part of the structure is the anchor, which simply serves as a way to hold VKOR within the proper orientation in the cell membrane such that all enzymatic components are in correct proximity for substrate binding and catalysis. | ||
| + | |||
=== Active Site === | === Active Site === | ||
| - | Within the four transmembrane helices lies the <scene name='90/906893/Active_site/4'>active site</scene>. The active site is comprised of a hydrophobic pocket containing two hydrophilic residues, | + | Within the four transmembrane helices lies the <scene name='90/906893/Active_site/4'>active site</scene>. The active site is comprised of a hydrophobic pocket containing two hydrophilic residues, N80 and Y139, that interact with substrates and ligands alike. The hydrophobic pocket provides specificity to the region while the hydrophilic residues have potential to hydrogen bond, allowing recognition and increasing specificity as well. Slightly above the active site is a crucial disulfide bridge that provides stabilization when a substrate is bound. This bridge occurs between C132 and C135, recurrent residues that continually aid in VKOR function. |
The active site plays a vital role in binding of any substrate or ligand to the VKOR. Upon binding, the VKOR will transition into a <scene name='90/906893/Closed_conformation/4'>closed conformation</scene> that will allow its catalytic mechanism to commence. | The active site plays a vital role in binding of any substrate or ligand to the VKOR. Upon binding, the VKOR will transition into a <scene name='90/906893/Closed_conformation/4'>closed conformation</scene> that will allow its catalytic mechanism to commence. | ||
| Line 38: | Line 41: | ||
This is a sample scene created with SAT to <scene name="/12/3456/Sample/1">color</scene> by Group, and another to make <scene name="/12/3456/Sample/2">a transparent representation</scene> of the protein. You can make your own scenes on SAT starting from scratch or loading and editing one of these sample scenes. | This is a sample scene created with SAT to <scene name="/12/3456/Sample/1">color</scene> by Group, and another to make <scene name="/12/3456/Sample/2">a transparent representation</scene> of the protein. You can make your own scenes on SAT starting from scratch or loading and editing one of these sample scenes. | ||
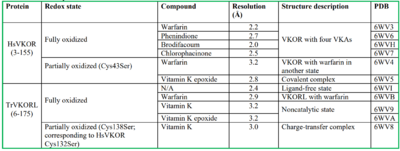
| + | [[Image:PDB table.png |400 px| right| thumb]] | ||
</StructureSection> | </StructureSection> | ||
| - | [[Image:PDB table.png |400 px| right| thumb]] | ||
== References == | == References == | ||
Revision as of 14:31, 13 April 2022
VKOR
| |||||||||||
References
1. Li, Weikai et al. “Structure of a bacterial homologue of vitamin K epoxide reductase.” Nature vol. 463,7280 (2010): 507-12. doi:10.1038/nature08720.
2. Liu S, Li S, Shen G, Sukumar N, Krezel AM, Li W. Structural basis of antagonizing the vitamin K catalytic cycle for anticoagulation. Science. 2021 Jan 1;371(6524):eabc5667. doi: 10.1126/science.abc5667. Epub 2020 Nov 5. PMID: 33154105; PMCID: PMC7946407.
- ↑ Hanson, R. M., Prilusky, J., Renjian, Z., Nakane, T. and Sussman, J. L. (2013), JSmol and the Next-Generation Web-Based Representation of 3D Molecular Structure as Applied to Proteopedia. Isr. J. Chem., 53:207-216. doi:http://dx.doi.org/10.1002/ijch.201300024
- ↑ Herraez A. Biomolecules in the computer: Jmol to the rescue. Biochem Mol Biol Educ. 2006 Jul;34(4):255-61. doi: 10.1002/bmb.2006.494034042644. PMID:21638687 doi:10.1002/bmb.2006.494034042644
- ↑ Unknown PubmedID 10.1126
- ↑ Unknown PubmedID 10.1126