This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Sandbox Reserved 1723
From Proteopedia
(Difference between revisions)
| Line 49: | Line 49: | ||
===== Cation ===== | ===== Cation ===== | ||
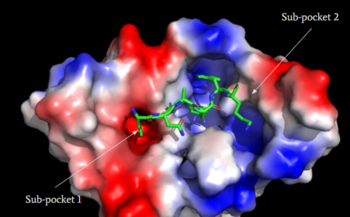
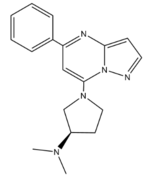
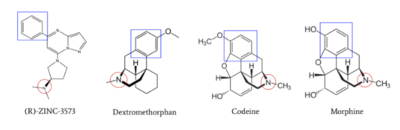
| - | <scene name='90/904328/Zinc/ | + | <scene name='90/904328/Zinc/3'>(R)-Zinc-3573</scene> is a cation ligand that selectively binds to MRGPRX2. Its N-dimethyl is inserted into subpocket 1 of the binding cavity with aromatic amino acid residues Phe-170, Trp-243, and Phe-244. In this <scene name='90/904328/Zizwithaa/3'>cavity</scene>, N-dimethyl group on the ligand makes ion pairs with Asp-184 and Glu-164. The ligand is then stabilized by stacking its ring with Trp-248 and the Cys-168 to Cys-180 disulfide bond <ref name="Yang">Yang, Fan, et al. "Structure, function and pharmacology of human itch receptor complexes." Nature, Nature Publishing Group, 17 November 2021, https://www.nature.com/articles/s41586-021-04077-y</ref>. |
===== Peptide ===== | ===== Peptide ===== | ||
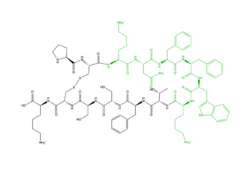
| - | <scene name='90/904328/Overview_x2_c_pt_3/ | + | <scene name='90/904328/Overview_x2_c_pt_3/4'>Cortistatin-14</scene> is one of the peptide ligands that binds to MRGPRX2. Cortistatin-14 interacts with the binding pocket through an <scene name='90/904328/Zic14_pt_3/4'>electrostatic</scene> interaction in sub-pocket 1 between Lys-3 on the peptide and Glu-164 and Asp-184 on MRGPRX2 <ref name="Cao"/>. Additionally, there are hydrophobic interactions in sub-pocket 2 between the peptide and the binding pocket due to the large hydrophobic amino acids on Cortistatin-14. |
[[Image:Newzinc.PNG |150px|center|thumb|'''Figure 3''': Structure of R-Zinc 3573. A cationic ligand selected for binding to MRGPRX2. This ligand was used as a probe for MRGPRX2 function and stabilization for structure determination. <ref name="Yang">Yang, Fan, et al. "Structure, function and pharmacology of human itch receptor complexes." Nature, Nature Publishing Group, 17 November 2021, https://www.nature.com/articles/s41586-021-04077-y</ref>]][[Image: Peptide.PNG|250px|center|thumb|'''Figure 4''': Structure of Cortistatin-14 with resolved amino acids highlighted in green.<ref name="Yang">Yang, Fan, et al. "Structure, function and pharmacology of human itch receptor complexes." Nature, Nature Publishing Group, 17 November 2021, https://www.nature.com/articles/s41586-021-04077-y</ref>]] | [[Image:Newzinc.PNG |150px|center|thumb|'''Figure 3''': Structure of R-Zinc 3573. A cationic ligand selected for binding to MRGPRX2. This ligand was used as a probe for MRGPRX2 function and stabilization for structure determination. <ref name="Yang">Yang, Fan, et al. "Structure, function and pharmacology of human itch receptor complexes." Nature, Nature Publishing Group, 17 November 2021, https://www.nature.com/articles/s41586-021-04077-y</ref>]][[Image: Peptide.PNG|250px|center|thumb|'''Figure 4''': Structure of Cortistatin-14 with resolved amino acids highlighted in green.<ref name="Yang">Yang, Fan, et al. "Structure, function and pharmacology of human itch receptor complexes." Nature, Nature Publishing Group, 17 November 2021, https://www.nature.com/articles/s41586-021-04077-y</ref>]] | ||
Revision as of 18:53, 18 April 2022
| This Sandbox is Reserved from February 28 through September 1, 2022 for use in the course CH462 Biochemistry II taught by R. Jeremy Johnson at the Butler University, Indianapolis, USA. This reservation includes Sandbox Reserved 1700 through Sandbox Reserved 1729. |
To get started:
More help: Help:Editing |
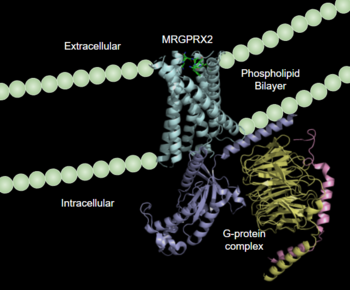
Human Itch GPCR
| |||||||||||
References
- ↑ 1.0 1.1 1.2 1.3 1.4 1.5 1.6 Cao, Can, et al. "Structure, function and pharmacology of human itch GPCRs." Nature, Nature Publishing Group, 17 November 2021, https://www.nature.com/articles/s41586-021-04126-6
- ↑ Thal, David M., et al. "Structural insights into G-protein-coupled receptor allostery." Nature, Nature Publishing Group, 04 July 2018, https://www.nature.com/articles/s41586-018-0259-z
- ↑ 3.0 3.1 Zhang D, Zhao Q, Wu B. Structural Studies of G Protein-Coupled Receptors. Mol Cells. 2015 Oct;38(10):836-42. doi: 10.14348/molcells.2015.0263. Epub 2015, Oct 15. PMID:26467290 doi:http://dx.doi.org/10.14348/molcells.2015.0263
- ↑ 4.0 4.1 4.2 Zhou Q, Yang D, Wu M, Guo Y, Guo W, Zhong L, Cai X, Dai A, Jang W, Shakhnovich EI, Liu ZJ, Stevens RC, Lambert NA, Babu MM, Wang MW, Zhao S. Common activation mechanism of class A GPCRs. Elife. 2019 Dec 19;8. pii: 50279. doi: 10.7554/eLife.50279. PMID:31855179 doi:http://dx.doi.org/10.7554/eLife.50279
- ↑ 5.0 5.1 5.2 5.3 5.4 5.5 5.6 Yang, Fan, et al. "Structure, function and pharmacology of human itch receptor complexes." Nature, Nature Publishing Group, 17 November 2021, https://www.nature.com/articles/s41586-021-04077-y
- ↑ 6.0 6.1 Schonegge, Anne-Marie, et al. "Evolutionary action and structural basis of the allosteric switch controlling β2AR functional selectivity." Nature, Nature Publishing Group, 18 December 2017, https://www.nature.com/articles/s41467-017-02257-x
- ↑ Sandoval, A., et al. "The Molecular Switching Mechanism at the Conserved D(E)RY Motif in Class-A GPCRs." Biophysical journal, 111(1), 79-89. https://doi.org/10.1016/j.bpj.2016.06.004
- ↑ Katritch V, Fenalti G, Abola EE, Roth BL, Cherezov V, Stevens RC. Allosteric sodium in class A GPCR signaling. Trends Biochem Sci. 2014 May;39(5):233-44. doi: 10.1016/j.tibs.2014.03.002. Epub , 2014 Apr 21. PMID:24767681 doi:http://dx.doi.org/10.1016/j.tibs.2014.03.002
- ↑ Babina, M., et al. "MRGPRX2 Is the Codeine Receptor of Human Skin Mast Cells: Desensitization through β-Arrestin and Lack of Correlation with the FcεRI Pathway." Journal of Investigative Dermatology, 141(6), 1286-1296. https://doi.org/10.1016/j.jid.2020.09.017