This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Sandbox Reserved 1767
From Proteopedia
| Line 42: | Line 42: | ||
=== Ras/Raf === | === Ras/Raf === | ||
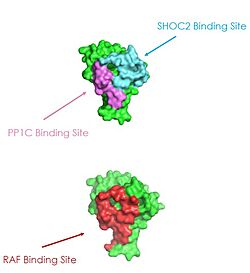
| - | [[Image:pic3.jpg|250 px|right|thumb|Figure 2: MRAS binding sites with SHOC2, PP1C, and RAF.<ref name="Liau">PMID: 35768504</ref>.]] | + | [[Image:pic3.jpg|250 px|right|thumb|Figure 2: MRAS binding sites with SHOC2, PP1C, and RAF (PDB 7DSO).<ref name="Liau">PMID: 35768504</ref>.]] |
| - | + | ||
| - | + | ||
| + | Ras proteins are GTP-dependent intracellular switches that are anchored to the plasma membrane. Ras proteins activate RAF kinases through direct binding and membrane recruitment, resulting in RAF dimerization and pathway activation. <ref name="Liau">PMID: 35768504</ref>. Ras has a hydrophobic fatty acid tail, keeping it anchored to the membrane. There are no known membrane interacting regions on SHOC2 and PP1C, meaning MRAS likely recruits them to the membrane. Significant amount of steric overlap is seen in MRAS for the binding sites of PP1C, SHOC2, and Raf. MRAS is shown in green, with the SHOC2 binding site colored cyan, the PP1C binding site colored green, and the RAF binding site shown in red on a different RAS protein. Hence, multiple Ras proteins are required for further activation of the receptor tyrosine kinase pathway. Due to the significant overlap in binding domains, one Ras molecule is needed to recruit SHOC2 and PP1C to the membrane, and one Ras molecule is needed activate Raf. The ability of Ras-GTP to cluster at the membrane is a crucial capability for this protein complex. This anchoring is possible due to the presence of a hydrophobic fatty acid tail on Ras. The presence of this palmitoyl tail on Raf also localizes the complex to the cell membrane, allowing only for 2D movement and increasing local concentrations of the players needed in this signaling pathway. | ||
== Structure of Active Site == | == Structure of Active Site == | ||
Revision as of 18:55, 11 April 2023
| This Sandbox is Reserved from February 27 through August 31, 2023 for use in the course CH462 Biochemistry II taught by R. Jeremy Johnson at the Butler University, Indianapolis, USA. This reservation includes Sandbox Reserved 1765 through Sandbox Reserved 1795. |
To get started:
More help: Help:Editing |
Contents |
SHOC2-PP1C-MRAS
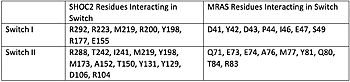
| |||||||||||
Protopedia Resources
References
- ↑ 1.0 1.1 1.2 1.3 1.4 1.5 Kwon JJ, Hajian B, Bian Y, Young LC, Amor AJ, Fuller JR, Fraley CV, Sykes AM, So J, Pan J, Baker L, Lee SJ, Wheeler DB, Mayhew DL, Persky NS, Yang X, Root DE, Barsotti AM, Stamford AW, Perry CK, Burgin A, McCormick F, Lemke CT, Hahn WC, Aguirre AJ. Structure-function analysis of the SHOC2-MRAS-PP1C holophosphatase complex. Nature. 2022 Jul 13. pii: 10.1038/s41586-022-04928-2. doi:, 10.1038/s41586-022-04928-2. PMID:35831509 doi:http://dx.doi.org/10.1038/s41586-022-04928-2
- ↑ 2.0 2.1 2.2 Lavoie H, Therrien M. Structural keys unlock RAS-MAPK cellular signalling pathway. Nature. 2022 Sep;609(7926):248-249. PMID:35970881 doi:10.1038/d41586-022-02189-7
- ↑ Hauseman ZJ, Fodor M, Dhembi A, Viscomi J, Egli D, Bleu M, Katz S, Park E, Jang DM, Porter KA, Meili F, Guo H, Kerr G, Molle S, Velez-Vega C, Beyer KS, Galli GG, Maira SM, Stams T, Clark K, Eck MJ, Tordella L, Thoma CR, King DA. Structure of the MRAS-SHOC2-PP1C phosphatase complex. Nature. 2022 Jul 13. pii: 10.1038/s41586-022-05086-1. doi:, 10.1038/s41586-022-05086-1. PMID:35830882 doi:http://dx.doi.org/10.1038/s41586-022-05086-1
- ↑ 4.0 4.1 4.2 4.3 4.4 Liau NPD, Johnson MC, Izadi S, Gerosa L, Hammel M, Bruning JM, Wendorff TJ, Phung W, Hymowitz SG, Sudhamsu J. Structural basis for SHOC2 modulation of RAS signalling. Nature. 2022 Jun 29. pii: 10.1038/s41586-022-04838-3. doi:, 10.1038/s41586-022-04838-3. PMID:35768504 doi:http://dx.doi.org/10.1038/s41586-022-04838-3
- ↑ 5.0 5.1 Young LC, Hartig N, Boned Del Río I, Sari S, Ringham-Terry B, Wainwright JR, Jones GG, McCormick F, Rodriguez-Viciana P. SHOC2-MRAS-PP1 complex positively regulates RAF activity and contributes to Noonan syndrome pathogenesis. Proc Natl Acad Sci U S A. 2018 Nov 6;115(45):E10576-E10585. PMID:30348783 doi:10.1073/pnas.1720352115
- ↑ 6.0 6.1 6.2 6.3 Hurley TD, Yang J, Zhang L, Goodwin KD, Zou Q, Cortese M, Dunker AK, DePaoli-Roach AA. Structural basis for regulation of protein phosphatase 1 by inhibitor-2. J Biol Chem. 2007 Sep 28;282(39):28874-83. Epub 2007 Jul 18. PMID:17636256 doi:http://dx.doi.org/10.1074/jbc.M703472200
1. Hauseman ZJ, Fodor M, Dhembi A, Viscomi J, Egli D, Bleu M, Katz S, Park E, Jang DM, Porter KA, Meili F, Guo H, Kerr G, Mollé S, Velez-Vega C, Beyer KS, Galli GG, Maira SM, Stams T, Clark K, Eck MJ, Tordella L, Thoma CR, King DA. Structure of the MRAS-SHOC2-PP1C phosphatase complex. Nature. 2022 Sep;609(7926):416-423. doi: 10.1038/s41586-022-05086-1. Epub 2022 Jul 13. PMID: 35830882; PMCID: PMC9452295.[1].
2. Hurley TD, Yang J, Zhang L, Goodwin KD, Zou Q, Cortese M, Dunker AK, DePaoli-Roach AA. Structural basis for regulation of protein phosphatase 1 by inhibitor-2. J Biol Chem. 2007 Sep 28;282(39):28874-28883. doi: 10.1074/jbc.M703472200. Epub 2007 Jul 18. PMID: 17636256.[2].
3. Kwon JJ, Hajian B, Bian Y, Young LC, Amor AJ, Fuller JR, Fraley CV, Sykes AM, So J, Pan J, Baker L, Lee SJ, Wheeler DB, Mayhew DL, Persky NS, Yang X, Root DE, Barsotti AM, Stamford AW, Perry CK, Burgin A, McCormick F, Lemke CT, Hahn WC, Aguirre AJ. Structure-function analysis of the SHOC2-MRAS-PP1C holophosphatase complex. Nature. 2022 Sep;609(7926):408-415. doi: 10.1038/s41586-022-04928-2. Epub 2022 Jul 13. PMID: 35831509; PMCID: PMC9694338.[3].
4. Liau NPD, Johnson MC, Izadi S, Gerosa L, Hammel M, Bruning JM, Wendorff TJ, Phung W, Hymowitz SG, Sudhamsu J. Structural basis for SHOC2 modulation of RAS signalling. Nature. 2022 Sep;609(7926):400-407. doi: 10.1038/s41586-022-04838-3. Epub 2022 Jun 29. PMID: 35768504; PMCID: PMC9452301.[4].
5. Lavoie H, Therrien M. Structural keys unlock RAS-MAPK cellular signalling pathway. Nature. 2022 Sep;609(7926):248-249. doi: 10.1038/d41586-022-02189-7. PMID: 35970881.[5].
6. Young LC, Hartig N, Boned Del Río I, Sari S, Ringham-Terry B, Wainwright JR, Jones GG, McCormick F, Rodriguez-Viciana P. SHOC2-MRAS-PP1 complex positively regulates RAF activity and contributes to Noonan syndrome pathogenesis. Proc Natl Acad Sci U S A. 2018 Nov 6;115(45):E10576-E10585. doi: 10.1073/pnas.1720352115. Epub 2018 Oct 22. PMID: 30348783; PMCID: PMC6233131.[6].
Student Contributors
- Sloan August
- Rosa Trippel
- Kayla Wilhoite