This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Doppel
From Proteopedia
| Line 1: | Line 1: | ||
| - | Doppel (Dpl), named for '''''do'''wnstream '''p'''rion '''p'''rotein like''<ref> | + | Doppel (Dpl), named for '''''do'''wnstream '''p'''rion '''p'''rotein-like''<ref>Moore, R ''et al.'' (1999) Ataxia in Prion Protein (PrP)-deficient mice is associated with upregulation of the novel PrP-like protein Doppel ''J. Mol. Biol.'' '''292''', 797-817</ref>, is a homolog of the [[prion protein]] (PrP). It is a cell surface glycoprotein. |
==Structure of Dpl== | ==Structure of Dpl== | ||
{{STRUCTURE_1lg4 | PDB=1lg4 | SCENE= }} | {{STRUCTURE_1lg4 | PDB=1lg4 | SCENE= }} | ||
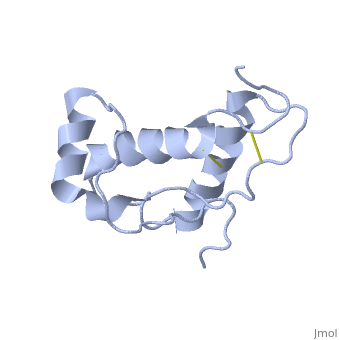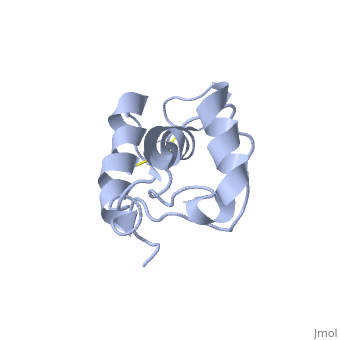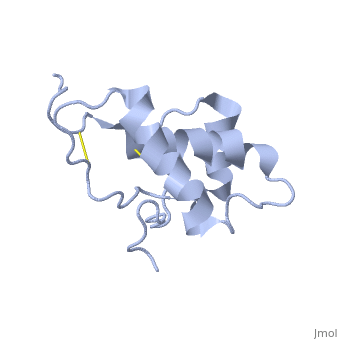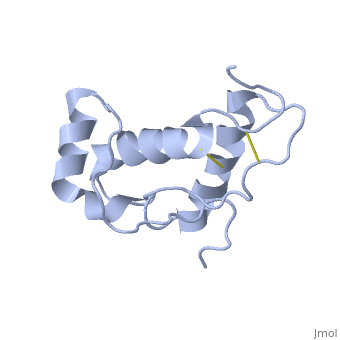
| - | Dpl has the same fold as PrP, with three alpha helices and two short beta strands, however it differs in that the third helix has a significant kink in it and it also contains two disulphide bonds. | + | Dpl has the same fold as PrP, with three alpha helices and two short beta strands<ref>Mo H ''et al.'' (2001) ''Proc. Natl. Acad. Sci. USA'' '''98''',2352-7</ref>. however it differs in that the third helix has a significant kink in it and it also contains two disulphide bonds. |
| + | |||
| + | The structure mutant PrP with the additional disulphide bond was also determoned <ref>Zahn R ''et al.'' (2003) NMR structure of a variant human prion protein with two disulfide bridges '' J. Mol. Biol.'' '''326''', 225-34. | ||
| + | |||
=Related structures= | =Related structures= | ||
| Line 14: | Line 17: | ||
=References= | =References= | ||
| - | + | </reference> | |
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
Revision as of 09:18, 15 December 2008
Doppel (Dpl), named for downstream prion protein-like[1], is a homolog of the prion protein (PrP). It is a cell surface glycoprotein.
Structure of Dpl
| |||||||||
| 1lg4, 20 NMR models () | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Gene: | Prnd (Homo sapiens) | ||||||||
| Related: | 1i17 | ||||||||
| |||||||||
| |||||||||
| |||||||||
| Resources: | FirstGlance, OCA, RCSB, PDBsum | ||||||||
| Coordinates: | save as pdb, mmCIF, xml | ||||||||
Dpl has the same fold as PrP, with three alpha helices and two short beta strands[2]. however it differs in that the third helix has a significant kink in it and it also contains two disulphide bonds.
The structure mutant PrP with the additional disulphide bond was also determoned [3]