This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Chaperonin
From Proteopedia
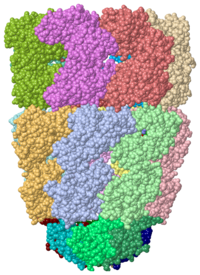
Chaperonins (CPN) are oligomeric proteins that mediate the folding of polypeptide chains. Group I CPN are found in bacteria, chloroplasts and mitochondria. For an introductory overview, see Chaperonins in Wikipedia.
The most characterized CPN are in the GroEL/GroES complex from Escherichia coli and CPN60/CPN10 from Thermus thermophilus. The larger subunit (GroEL, CPN60) contains 3 domains. The apical domain is the one which binds the substrate. Group II CPNs are found in eukaryotic cytosol and archaea. Thermosome is a CPN complex found in archaea. CCT is a CPN complex found in eukarya.
3D Structures of Chaperonin
Updated on 20-November-2014
Proteopedia Page Contributors and Editors (what is this?)
Michal Harel, Alexander Berchansky, Joel L. Sussman, Eric Martz, Jaime Prilusky
