This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Sandbox Reserved 930
From Proteopedia
| This Sandbox is Reserved from 01/04/2014, through 30/06/2014 for use in the course "510042. Protein structure, function and folding" taught by Prof Adrian Goldman, Tommi Kajander, Taru Meri, Konstantin Kogan and Juho Kellosalo at the University of Helsinki. This reservation includes Sandbox Reserved 923 through Sandbox Reserved 947. |
To get started:
More help: Help:Editing |
Contents |
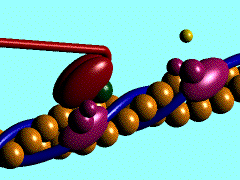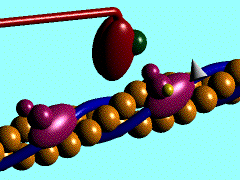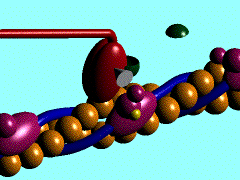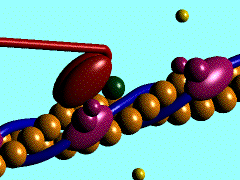
Scallop myosin head in its pre power stroke state
Introduction
Muscle contraction is achieved by the sliding of myosin filament (thick filament) and actin filament (thin filament). As the major constituent of myosin filament, a myosin molecule is a multifunctional protein, which has a ATP catalytic site and a actin binding site on each of the globular subfragment-1 unit. With the presence of Ca and Mg, ATP ís able to be hydrolyzed and triggers following myosin-actin detachment, reattachment and power stroke. This process transformed chemical energy to mechanical force.
Myosin subfragment-1 with MgADP
|
Mg
ADP
Section 3
This is the structure for to show. sdfbbsjfjsbfskjbf sjdfnjldns. IJI RR.
Conclusions
</StructureSection>