This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
JMS/Sandbox/trs
From Proteopedia
Contents |
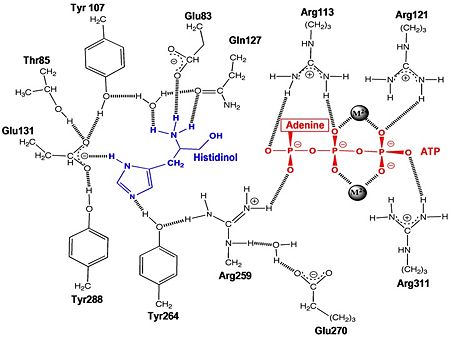
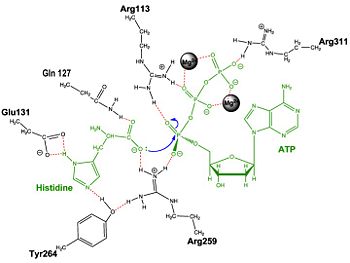
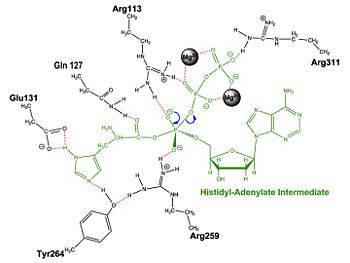
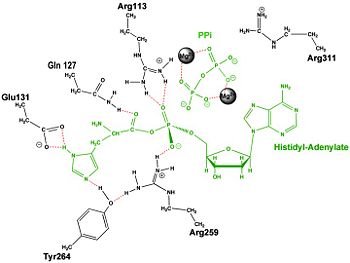
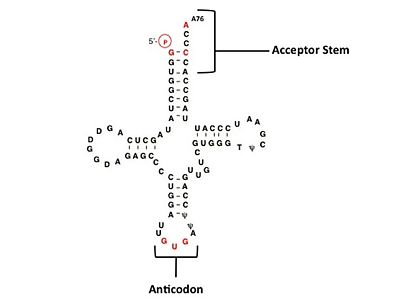
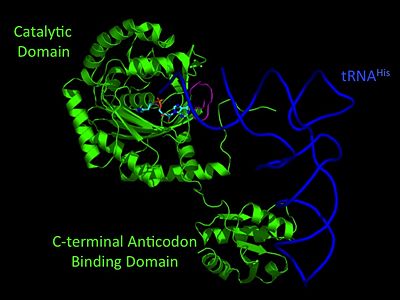
Histidyl-tRNA Synthetase
| |||||||||||
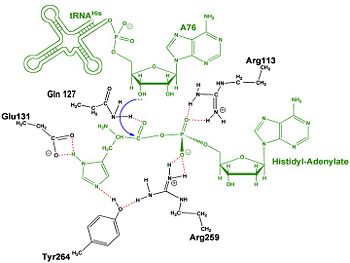
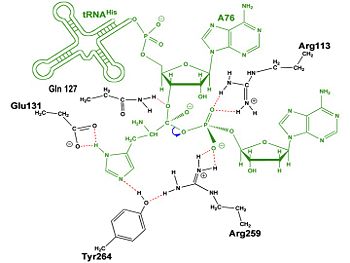
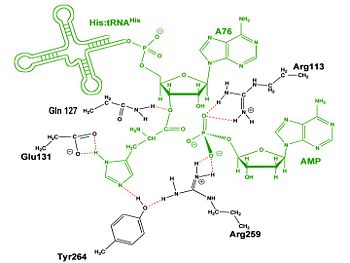
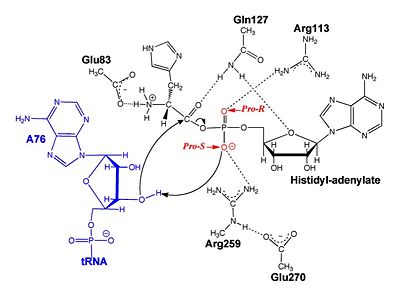
Mechanism of the Adenylation Reaction
| |||||||||||
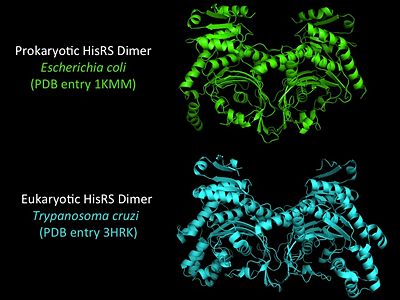
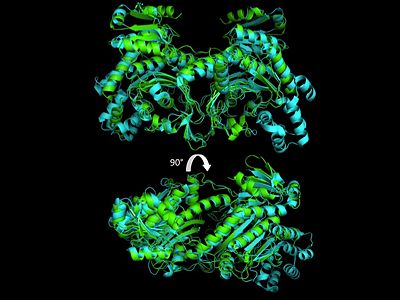
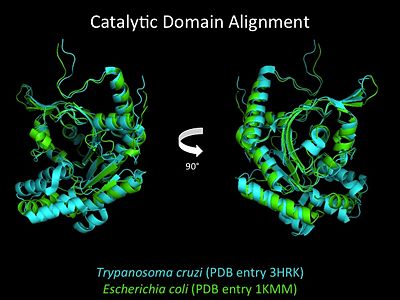
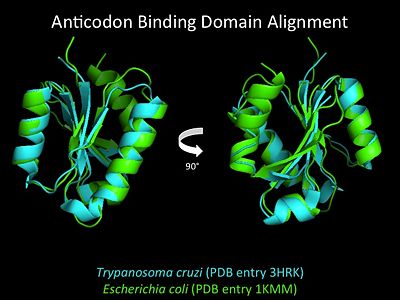
3D Structures of Histidyl-tRNA Synthetase
Bacteria
Eukaryota
Archara
References
- ↑ 1.0 1.1 1.2 Eriani G, Delarue M, Poch O, Gangloff J, Moras D. Partition of tRNA synthetases into two classes based on mutually exclusive sets of sequence motifs. Nature. 1990 Sep 13;347(6289):203-6. PMID:2203971 doi:http://dx.doi.org/10.1038/347203a0
- ↑ Cusack S, Hartlein M, Leberman R. Sequence, structural and evolutionary relationships between class 2 aminoacyl-tRNA synthetases. Nucleic Acids Res. 1991 Jul 11;19(13):3489-98. PMID:1852601
- ↑ 3.0 3.1 3.2 3.3 3.4 3.5 Francklyn, C., and Arnez, J.G. (2004) in Aminoacyl-tRNA Synthetases (Ibba, M.,Francklyn, C.,Cusack, S.. Eds.) Landes Publishing, Austin, TX
- ↑ 4.0 4.1 4.2 4.3 4.4 4.5 4.6 4.7 Arnez JG, Augustine JG, Moras D, Francklyn CS. The first step of aminoacylation at the atomic level in histidyl-tRNA synthetase. Proc Natl Acad Sci U S A. 1997 Jul 8;94(14):7144-9. PMID:9207058
- ↑ Arnez JG, Harris DC, Mitschler A, Rees B, Francklyn CS, Moras D. Crystal structure of histidyl-tRNA synthetase from Escherichia coli complexed with histidyl-adenylate. EMBO J. 1995 Sep 1;14(17):4143-55. PMID:7556055
- ↑ Arnez JG, Moras D. Structural and functional considerations of the aminoacylation reaction. Trends Biochem Sci. 1997 Jun;22(6):211-6. PMID:9204708
- ↑ 7.0 7.1 Belrhali H, Yaremchuk A, Tukalo M, Berthet-Colominas C, Rasmussen B, Bosecke P, Diat O, Cusack S. The structural basis for seryl-adenylate and Ap4A synthesis by seryl-tRNA synthetase. Structure. 1995 Apr 15;3(4):341-52. PMID:7613865
- ↑ Arnez JG, Flanagan K, Moras D, Simonson T. Engineering an Mg2+ site to replace a structurally conserved arginine in the catalytic center of histidyl-tRNA synthetase by computer experiments. Proteins. 1998 Aug 15;32(3):362-80. PMID:9715912
- ↑ Ruhlmann A, Cramer F, Englisch U. Isolation and analysis of mutated histidyl-tRNA synthetases from Escherichia coli. Biochem Biophys Res Commun. 1997 Aug 8;237(1):192-201. PMID:9266856 doi:10.1006/bbrc.1997.7108
- ↑ Poterszman A, Delarue M, Thierry JC, Moras D. Synthesis and recognition of aspartyl-adenylate by Thermus thermophilus aspartyl-tRNA synthetase. J Mol Biol. 1994 Nov 25;244(2):158-67. PMID:7966328 doi:http://dx.doi.org/10.1006/jmbi.1994.1716
- ↑ 11.0 11.1 11.2 11.3 11.4 11.5 11.6 Guth E, Connolly SH, Bovee M, Francklyn CS. A substrate-assisted concerted mechanism for aminoacylation by a class II aminoacyl-tRNA synthetase. Biochemistry. 2005 Mar 15;44(10):3785-94. PMID:15751955 doi:10.1021/bi047923h
- ↑ 12.0 12.1 12.2 12.3 Guth EC, Francklyn CS. Kinetic discrimination of tRNA identity by the conserved motif 2 loop of a class II aminoacyl-tRNA synthetase. Mol Cell. 2007 Feb 23;25(4):531-42. PMID:17317626 doi:10.1016/j.molcel.2007.01.015
- ↑ Moulinier L, Eiler S, Eriani G, Gangloff J, Thierry JC, Gabriel K, McClain WH, Moras D. The structure of an AspRS-tRNA(Asp) complex reveals a tRNA-dependent control mechanism. EMBO J. 2001 Sep 17;20(18):5290-301. PMID:11566892 doi:http://dx.doi.org/10.1093/emboj/20.18.5290
- ↑ Cavarelli J, Moras D. Recognition of tRNAs by aminoacyl-tRNA synthetases. FASEB J. 1993 Jan;7(1):79-86. PMID:8422978
- ↑ Rould MA, Perona JJ, Steitz TA. Structural basis of anticodon loop recognition by glutaminyl-tRNA synthetase. Nature. 1991 Jul 18;352(6332):213-8. PMID:1857417 doi:http://dx.doi.org/10.1038/352213a0
- ↑ Muramatsu T, Nishikawa K, Nemoto F, Kuchino Y, Nishimura S, Miyazawa T, Yokoyama S. Codon and amino-acid specificities of a transfer RNA are both converted by a single post-transcriptional modification. Nature. 1988 Nov 10;336(6195):179-81. PMID:3054566 doi:http://dx.doi.org/10.1038/336179a0
- ↑ Fromant M, Plateau P, Blanquet S. Function of the extra 5'-phosphate carried by histidine tRNA. Biochemistry. 2000 Apr 11;39(14):4062-7. PMID:10747795
- ↑ Himeno H, Hasegawa T, Ueda T, Watanabe K, Miura K, Shimizu M. Role of the extra G-C pair at the end of the acceptor stem of tRNA(His) in aminoacylation. Nucleic Acids Res. 1989 Oct 11;17(19):7855-63. PMID:2678006
- ↑ Yan W, Francklyn C. tRNA selection by a class II aminoacyl-tRNA synthetase: the role of accessory domains and inter-domain communication in RNA recognition. Nucleic Acids Symp Ser. 1995;(33):167-9. PMID:8643360
- ↑ Connolly SA, Rosen AE, Musier-Forsyth K, Francklyn CS. G-1:C73 recognition by an arginine cluster in the active site of Escherichia coli histidyl-tRNA synthetase. Biochemistry. 2004 Feb 3;43(4):962-9. PMID:14744140 doi:10.1021/bi035708f
- ↑ Merritt EA, Arakaki TL, Gillespie JR, Larson ET, Kelley A, Mueller N, Napuli AJ, Kim J, Zhang L, Verlinde CL, Fan E, Zucker F, Buckner FS, van Voorhis WC, Hol WG. Crystal structures of trypanosomal histidyl-tRNA synthetase illuminate differences between eukaryotic and prokaryotic homologs. J Mol Biol. 2010 Mar 26;397(2):481-94. Epub 2010 Feb 2. PMID:20132829 doi:10.1016/j.jmb.2010.01.051
- ↑ Brindefalk B, Viklund J, Larsson D, Thollesson M, Andersson SG. Origin and evolution of the mitochondrial aminoacyl-tRNA synthetases. Mol Biol Evol. 2007 Mar;24(3):743-56. Epub 2006 Dec 20. PMID:17182897 doi:10.1093/molbev/msl202
- ↑ Merritt EA, Arakaki TL, Gillespie JR, Larson ET, Kelley A, Mueller N, Napuli AJ, Kim J, Zhang L, Verlinde CL, Fan E, Zucker F, Buckner FS, van Voorhis WC, Hol WG. Crystal structures of trypanosomal histidyl-tRNA synthetase illuminate differences between eukaryotic and prokaryotic homologs. J Mol Biol. 2010 Mar 26;397(2):481-94. Epub 2010 Feb 2. PMID:20132829 doi:10.1016/j.jmb.2010.01.051
- ↑ O'Hanlon TP, Miller FW. Genomic organization, transcriptional mapping, and evolutionary implications of the human bi-directional histidyl-tRNA synthetase locus (HARS/HARSL). Biochem Biophys Res Commun. 2002 Jun 14;294(3):609-14. PMID:12056811 doi:10.1016/S0006-291X(02)00525-9
- ↑ O'Hanlon TP, Raben N, Miller FW. A novel gene oriented in a head-to-head configuration with the human histidyl-tRNA synthetase (HRS) gene encodes an mRNA that predicts a polypeptide homologous to HRS. Biochem Biophys Res Commun. 1995 May 16;210(2):556-66. PMID:7755634