This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
1w06
From Proteopedia
|
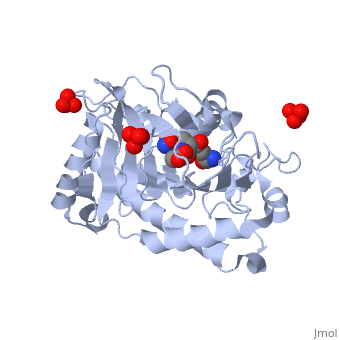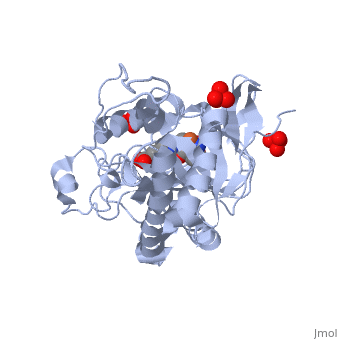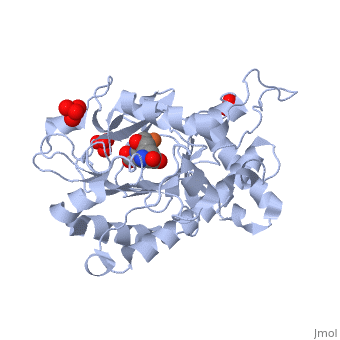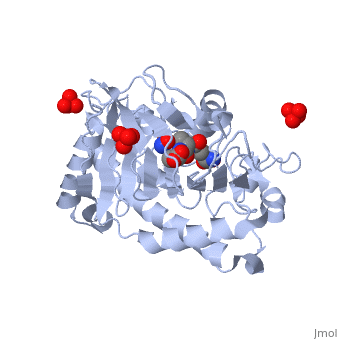
ISOPENICILLIN N SYNTHASE AMINOADIPOYL-CYSTEINYL-ALANINE-FE NO COMPLEX
Overview
Isopenicillin N synthase (IPNS), a non-heme iron(II)-dependent oxidase, catalyzes conversion of the tripeptide, delta-(l-alpha-aminoadipoyl)-l-cysteinyl-d-valine (ACV) to bicyclic, isopenicillin N (IPN), concomitant with the reduction of dioxygen to two, molecules of water. Incubation of the "truncated"substrate analogues, delta-(l-alpha-aminoadipoyl)-l-cysteinyl-glycine (ACG) and, delta-(l-alpha-aminoadipoyl)-l-cysteinyl-d-alanine (ACA) with IPNS has, previously been shown to afford acyclic products, in which the substrate, cysteinyl residue has undergone a two-electron oxidation. We report X-ray, crystal structures for the anaerobic IPNS/Fe(II)/ACG and IPNS/Fe(II)/ACA, complexes, both in the absence and presence of the dioxygen analogue, nitric oxide. The overall protein structures are very similar to those of, the corresponding IPNS/Fe(II)/ACV complexes; however, significant, differences are apparent in the vicinity of the active site iron. The, structure of the IPNS/Fe(II)/ACG complex reveals that the C-terminal, carboxylate of this substrate is oriented toward the active site iron, atom, apparently hydrogen-bonded to an additional water ligand at the, metal; this is a different binding mode to that observed in the, IPNS/Fe(II)/ACV complex. ACA binds to the metal in a manner that is, intermediate between those observed for ACV and ACG. The addition of NO to, these complexes initiates conformational changes such that both the, IPNS/Fe(II)/ACG/NO and IPNS/Fe(II)/ACA/NO structures closely resemble the, IPNS/Fe(II)/ACV/NO complex. These results further demonstrate the, feasibility of metal-centered rearrangements in catalysis by non-heme iron, enzymes and provide insight into the delicate balance between, hydrophilic-hydrophobic interactions and steric effects in the IPNS active, site.
About this Structure
1W06 is a Single protein structure of sequence from Emericella nidulans with FE2, SO4, W05 and NO as ligands. Active as Isopenicillin-N synthase, with EC number 1.21.3.1 Structure known Active Site: AC1. Full crystallographic information is available from OCA.
Reference
Structural studies on the reaction of isopenicillin N synthase with the truncated substrate analogues delta-(L-alpha-aminoadipoyl)-L-cysteinyl-glycine and delta-(L-alpha-aminoadipoyl)-L-cysteinyl-D-alanine., Long AJ, Clifton IJ, Roach PL, Baldwin JE, Rutledge PJ, Schofield CJ, Biochemistry. 2005 May 3;44(17):6619-28. PMID:15850395
Page seeded by OCA on Mon Nov 5 17:21:15 2007