This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Sandbox Reserved 1506
From Proteopedia
|
|
| This Sandbox is Reserved from 06/12/2018, through 30/06/2019 for use in the course "Structural Biology" taught by Bruno Kieffer at the University of Strasbourg, ESBS. This reservation includes Sandbox Reserved 1480 through Sandbox Reserved 1543. |
To get started:
More help: Help:Editing |
This is a default text for your page b6. Click above on edit this page to modify. Be careful with the < and > signs.
You may include any references to papers as in: the use of JSmol in Proteopedia [1] or to the article describing Jmol [2] to the rescue.
Function
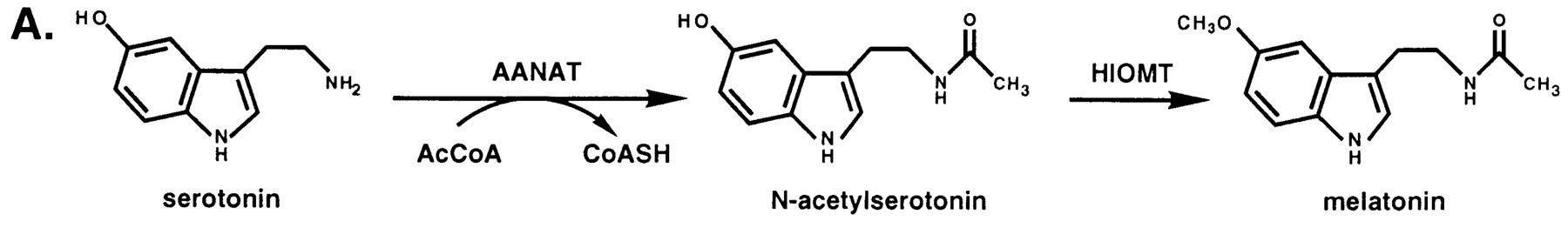
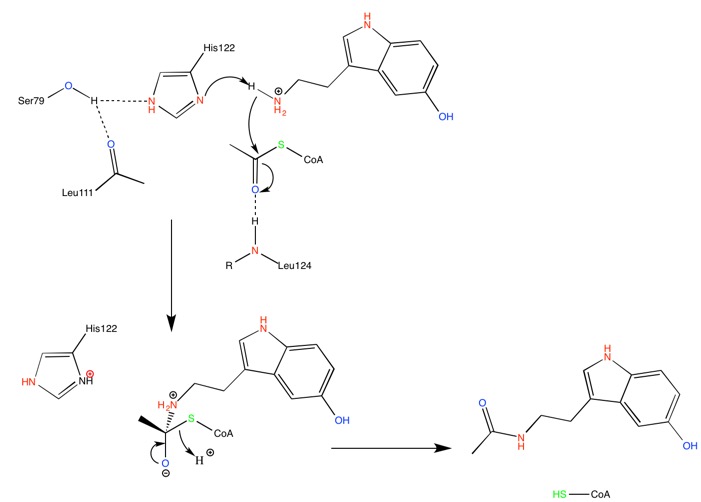
Serotonin N-acetyltransferase is also named aralkylamine-N-acetyltransferase (ANAAT). AANAT is a member of a large superfamily of proteins, referred to alternatively as the motif A/B or the GCN-5-related N-acetyltransferase (or GNAT) family. The role of these enzymes is to catalyse the acetylation all sorts of residues. ANAAT catalyzes the acetylation of the amine group on serotonin, an intermediate in melatonin synthesis. It is the penultimate enzyme in the melatonin pathway, as it is shown on fig1.[3].
Relevance
Circulating melatonin plays a role in the circadian rythm. Day/night differences in circulating melatonin levels provide a hormonal analog signal of environmental lighting, which is used in a variety of ways to optimize circadian and circannual rhythms in physiology[4]
Regulation
</StructureSection>
Cite error: Invalid <references> tag;
no input is allowed. Use <references />