This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Sandbox Reserved 1788
From Proteopedia
This page, as it appeared on June 14, 2016, was featured in this article in the journal Biochemistry and Molecular Biology Education.
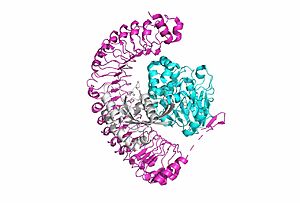
SHOC2-PP1C-MRAS
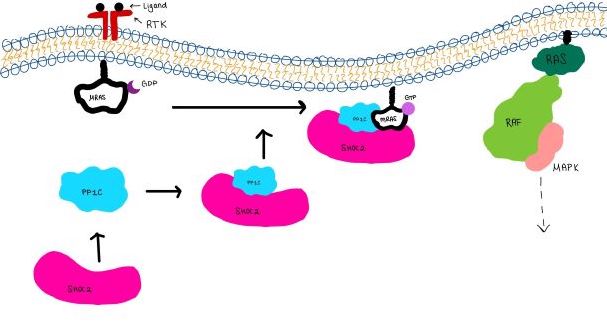
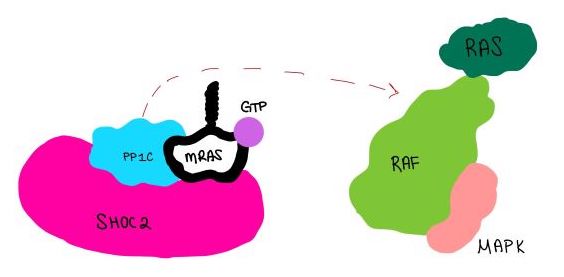
Introduction(SMP) is a ternary holoposphatase complex formed by the individual proteins: SHOC2, PP1C, and MRAS. Formation of this complex begins with a signal binding to a receptor tyrosine kinase receptor(RTK). This causes membrane-bound MRAS to exchange GDP for GTP. From here the complex comes together in the plasma membrane. Its role in MAPK signaling is the dephosphorylation of the RAF complex leading to further downstream signaling effects.
Overall StructureSHOC2is a crescent-shaped scaffold protein that is composed of 20 leucine-rich repeat domains that form a solenoid structure. The leucine-rich region forms a concave hydrophobic core which is necessary for binding with PP1C and MRAS. PP1Cis a catalytic protein. After forming a ternary complex, the on the protein interacts with Raf to act as a phosphatase and dephosphorylate Ser 259. MRASis a membrane bound structure that aids the complex in localizing near other structures such as the RAS-RAF-MAPK complex in order to initiate downstream signaling. In its inactive state, MRAS is bound to GDP. When signaled by growth factors, the GDP is exchanged for GTP. The now undergoes a conformational change of the . This conformational change activates the protein allowing it to bind more easily with the SHOC2-PP1C complex. In comparison to other RAS proteins, MRAS has a greater affinity for the SHOC2-PP1C complex. Key Ligand Interactions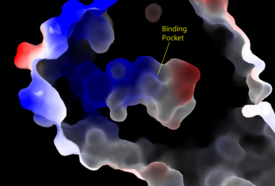 Figure 3: Electrostatic illustration of the amphipathic binding pocket of the LPA1 receptor. This binding pocket was revealed by cutting away the exterior or the protein. This binding pocket, located in the interior of the protein, has both polar and nonpolar regions. The blue and red coloration highlight the positively and negatively charged regions, respectively, and the white color shows the nonpolar region of the binding pocket. SHOC2 and PP1Con its leucine rich region(LRR). Specifically, on two broad surfaces between LRR2 and LRR5 and between LRR7 and LRR11. Mutations made to the LRR were shown to completely inhibit the binding of PP1C. Five main are made: E56-R182, E167-R203, E54-K180, R187-H178, R188-E155. The binding regions can also be shown as acidic and basic patches on and . The corresponding patches interact to form a . These interactions do not result in significant conformational changes.=== SHOC2 and MRAS === MRAS is initially bound to GDP causing it to be in its inactive state. This form cannot bind to the SHOC2-PP1C complex due to steric clashing. Once GDP is exchanged for GTP to activate the protein, occur within the switch I and switch II regions to allow . These include hydrogen bonds and pi stacking. The primary hydrogen bonds are R288-Q71 and R177-E47. Pi staking occurs at R104-R83. PP1C and MRASThe interactions between PP1C and MRAS are mediated by four main hydrogen bonds: R188-D48, M190-Q35, D197-H53, Q198-K36. It is unclear whether PP1C must bind to SHOC2 before MRAS binds or if PP1C and MRAS can bind to SHOC2 at the same time. Signaling Pathway
Disease RelevanceRASopathiesRASopathy is a broad term used to describe developmental syndromes that stem from germline mutations of proteins along the RAS/MAPK pathway such as SHOC2, PP1C, and MRAS. These mutations can be either gain or loss of function. Rasopathies can also lead to cancer. CancerBecause the RAS/MAPK pathway activated by SMP regulates cell proliferation and survival, overactivity can cause tumor formation and cancer. For example, the complex has been found to play a role in the perpetuation of melanoma, leukemia, and lung cancer. Future StudiesFurther study of the SMP complex includes clarification of the steps of the pathway. Firstly, the order of binding to form the SMP complex is unclear. Furthermore, the interaction between SMP and the Raf complex is largely unknown. Study into this step is especially important to understand how SMP activates downstream signaling. The current knowledge of SMP can be used to study possible treatments for rasopathies and cancer. For example, the development of inhibitors that target SMP binding could prevent the effects caused by mutations that overactivate SMP. Another possible point of inhibition is the growth factor that signals SHOC2-PP1C and Raf to the cell membrane.
References
Student ContributorsMadeline Gilbert Inaya Patel Rushda Hussein | ||||||||||||