This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Amyloid beta
From Proteopedia
(Difference between revisions)
| Line 1: | Line 1: | ||
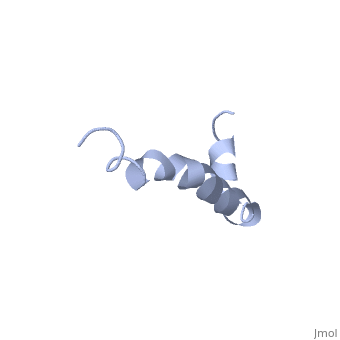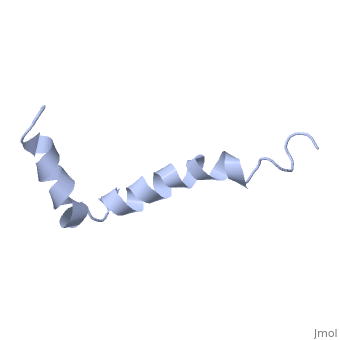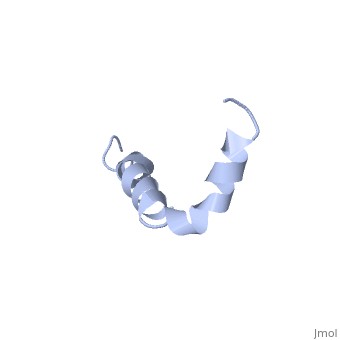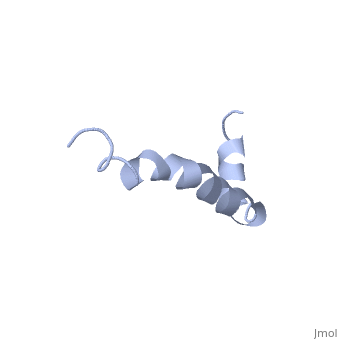
<StructureSection load=1iyt size='500' side='right' caption='amyloid-beta(1-42)', ([[1dm0]])' scene=''> | <StructureSection load=1iyt size='500' side='right' caption='amyloid-beta(1-42)', ([[1dm0]])' scene=''> | ||
==Introduction== | ==Introduction== | ||
| - | '''Alzheimer's disease''' is characterized by extracellular proteic plaques and intracellular neurofibrillary tangles.<ref name="structure">PMID: 12423364</ref> These plaques are collections of beta-amyloid <scene name='Amyloid_beta/Fibrils/1'>fibrils</scene> of aggregated beta-sheets. When amyloid beta comes into contact with metal ions and oxygen the result is the production of reactive oxygen species (especially hydrogen peroxide).<ref name="alz">PMID:18305836</ref> Amyloid beta also maintains the capacity to to induce pore formation in neuronal and endothelial cells which can trigger cell death.<ref name="structure" /><ref name="alz" /> Yet another source of amyloid beta toxicity stems from its ability to induce endothelial cell damage through the production of superoxide, though the mechanism of such induction is unclear.<ref name="alz" /> | + | '''Alzheimer's disease''' is characterized by extracellular proteic plaques and intracellular neurofibrillary tangles.<ref name="structure">PMID: 12423364</ref> These plaques are collections of beta-amyloid <scene name='Amyloid_beta/Fibrils/1'>fibrils</scene> of aggregated beta-sheets. When amyloid beta comes into contact with metal ions and oxygen the result is the production of reactive oxygen species (especially hydrogen peroxide).<ref name="alz">PMID:18305836</ref> Amyloid beta also maintains the capacity to to induce pore formation in neuronal and endothelial cells which can trigger cell death.<ref name="structure" /><ref name="alz" /> Yet another source of amyloid beta toxicity stems from its ability to induce endothelial cell damage through the production of superoxide, though the mechanism of such induction is unclear.<ref name="alz" /> While the presence of the fibril plaques remains a marker for Alzheimer's disease recent studies have suggested that alymoild beta oligomers more devestating effect occurs when it impairs long-term potentiation which decreases dendritic spine density in the hippocampal brain and impairs memory.<ref name="recent">PMID:22114742</ref> |
==Structure== | ==Structure== | ||
Revision as of 00:44, 29 November 2011
| |||||||||||