This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Amyloid beta
From Proteopedia
(Difference between revisions)
| Line 4: | Line 4: | ||
==Structure== | ==Structure== | ||
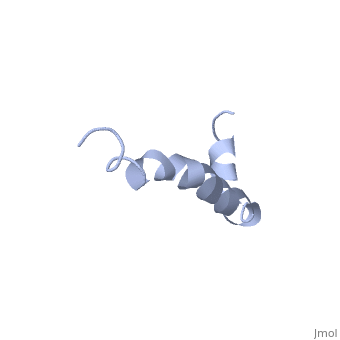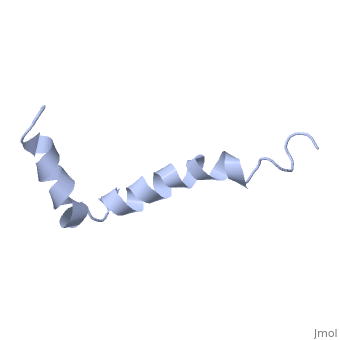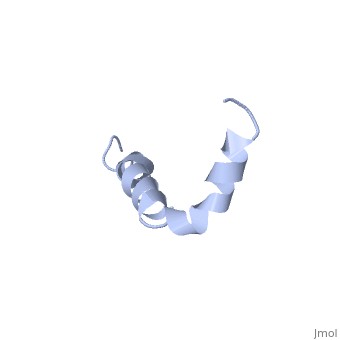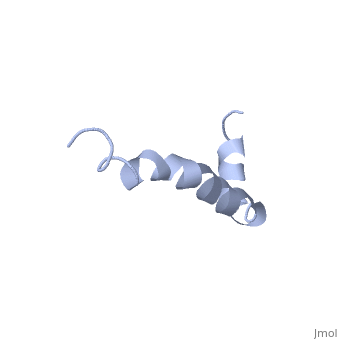
| - | Amyoloid beta is actually the <scene name='Amyloid_beta/C-term/1'>C-terminal</scene> of the <scene name='Amyloid_beta/App/1'>Amyloid Precursor Protein</scene> which is a type I membrane-spanning glycoprotein [[http://proteopedia.org/wiki/index.php/Amyloid_precursor_protein APP]].<ref name="alz" /> | + | Amyoloid beta is actually the <scene name='Amyloid_beta/C-term/1'>C-terminal</scene> of the <scene name='Amyloid_beta/App/1'>Amyloid Precursor Protein</scene> which is a type I membrane-spanning glycoprotein encoded on chromosome 21 [[http://proteopedia.org/wiki/index.php/Amyloid_precursor_protein APP]].<ref name="alz" /> Amyloid beta results from an abnormal cleavage by beta-secretase at the N-terminal and gamma-secretase at the C-terminal. The cleavage is nonspecific and results in peptides 39-43 amino acids in length, with 42 being the most common. Such cleavages occur most commonly in the plasma membrane is also occurs in neuronal membranes.<ref name="alz" /> |
| + | |||
The most reasonable structure determined structure consists of <scene name='Amyloid_beta/Two_helices/1'>two helices</scene>; the first helix (residues 8-25) is well defined and has an RMSD of 0.38 angstroms and the second (residues 28-38) is interrupted at the Ile32-Gly33 connection. The two helices are connected by a <scene name='Amyloid_beta/Kink/1'>kink</scene> (residues 26 and 27).<ref name="structure" /> | The most reasonable structure determined structure consists of <scene name='Amyloid_beta/Two_helices/1'>two helices</scene>; the first helix (residues 8-25) is well defined and has an RMSD of 0.38 angstroms and the second (residues 28-38) is interrupted at the Ile32-Gly33 connection. The two helices are connected by a <scene name='Amyloid_beta/Kink/1'>kink</scene> (residues 26 and 27).<ref name="structure" /> | ||
==References== | ==References== | ||
<references/> | <references/> | ||
Revision as of 06:16, 26 November 2011
| |||||||||||