This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Sandbox reserved 659
From Proteopedia
| Line 2: | Line 2: | ||
[[Image:1b8e_bio_r_500.jpg|thumb|left|260px|]] | [[Image:1b8e_bio_r_500.jpg|thumb|left|260px|]] | ||
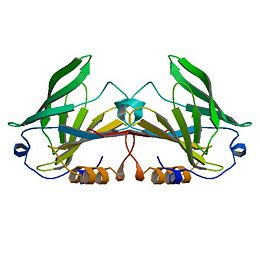
| - | [[β-Lactoglobulin]](β-LG) is the | + | [[β-Lactoglobulin]]β-Lactoglobulin(β-LG) is the bulk constituent of whey protein in most ruminant species and is also present in the milks of most mammalian species, although not in rodent, human, and lagomorph milks. It has been studied extensively for some time ( Tilley, 1960; Lyster, 1972; Kinsella and Whitehead, 1987; Hambling et al., 1992; Sawyer, 2003). β-LG is a lipocalin as is seen through its amino-acid sequence and structure (Flower et al., 2000). Lipocalins are a varied family which bind small hydrophobic ligands, and often act as transporters in vivo. The purpose of β-LG, however, is not clear. Genetic variants of β-LG expressing genes have been reported, especially in cows and several nonruminant species (Kontopidis, 2004). |
| - | + | ||
| - | + | ||
<StructureSection load='1B8E' size='350' side='right' caption='Structure of Beta Lactoglobulin (PDB entry [[1B8E]])' scene=''> | <StructureSection load='1B8E' size='350' side='right' caption='Structure of Beta Lactoglobulin (PDB entry [[1B8E]])' scene=''> | ||
Revision as of 14:24, 27 November 2012
Beta Lactoglobulin
β-Lactoglobulinβ-Lactoglobulin(β-LG) is the bulk constituent of whey protein in most ruminant species and is also present in the milks of most mammalian species, although not in rodent, human, and lagomorph milks. It has been studied extensively for some time ( Tilley, 1960; Lyster, 1972; Kinsella and Whitehead, 1987; Hambling et al., 1992; Sawyer, 2003). β-LG is a lipocalin as is seen through its amino-acid sequence and structure (Flower et al., 2000). Lipocalins are a varied family which bind small hydrophobic ligands, and often act as transporters in vivo. The purpose of β-LG, however, is not clear. Genetic variants of β-LG expressing genes have been reported, especially in cows and several nonruminant species (Kontopidis, 2004).
| |||||||||||
Physiological Function of β-LG
| |||||||||||
Tanford Transition of β-LG
| |||||||||||